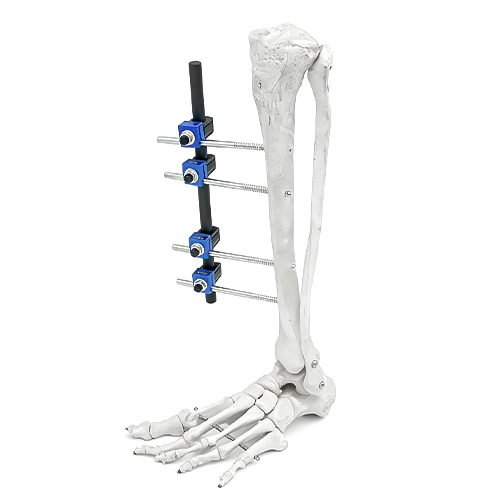
-
• Surgical Technique Videos: Monolateral leh Ilizarov frame inzawmkhawmna atana step by step-a hmuh theiha kaihhruaina.
• Operation Manuals (IFU): Instrument hman dan tur leh sterilization protocol chipchiar tak.
• On-site Support: Kan services (PDF P38)-a kan sawi tawh angin, distributor pressure tih tlem nan technical exchange meeting kan buatsaih a, hospital surgery follow-up kan support bawk.
Please Choose Your Language
-
English
-
Русский
-
简体中文
-
繁體中文
-
العربية
-
Français
-
Español
-
Português
-
Deutsch
-
italiano
-
日本語
-
한국어
-
Nederlands
-
Tiếng Việt
-
ไทย
-
Polski
-
Türkçe
-
አማርኛ
-
ພາສາລາວ
-
ភាសាខ្មែរ
-
Bahasa Melayu
-
ဗမာစာ
-
தமிழ்
-
Filipino
-
Bahasa Indonesia
-
magyar
-
Română
-
Čeština
-
Монгол
-
қазақ
-
Српски
-
हिन्दी
-
فارسی
-
Kiswahili
-
Slovenčina
-
Slovenščina
-
Norsk
-
Svenska
-
українська
-
Ελληνικά
-
Suomi
-
Հայերեն
-
עברית
-
Latine
-
Dansk
-
اردو
-
Shqip
-
বাংলা
-
Hrvatski
-
Afrikaans
-
Gaeilge
-
Eesti keel
-
Māori
-
සිංහල
-
नेपाली
-
Oʻzbekcha
-
latviešu
-
অসমীয়া
-
Aymara
-
Azərbaycan dili
-
Bamanankan
-
Euskara
-
Беларуская мова
-
भोजपुरी
-
Bosanski
-
Български
-
Català
-
Cebuano
-
Corsu
-
ދިވެހި
-
डोग्रिड ने दी
-
Esperanto
-
Eʋegbe
-
Frysk
-
Galego
-
ქართული
-
guarani
-
ગુજરાતી
-
Kreyòl ayisyen
-
Hausa
-
ʻŌlelo Hawaiʻi
-
Hmoob
-
íslenska
-
Igbo
-
Ilocano
-
Basa Jawa
-
ಕನ್ನಡ
-
Kinyarwanda
-
गोंगेन हें नांव
-
Krio we dɛn kɔl Krio
-
Kurdî
-
Kurdî
-
Кыргызча
-
Lingala
-
Lietuvių
-
Oluganda
-
Lëtzebuergesch
-
Македонски
-
मैथिली
-
Malagasy
-
മലയാളം
-
Malti
-
मराठी
-
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
-
Mizo tawng
-
Chichewa
-
ଓଡ଼ିଆ
-
Afaan Oromoo
-
پښتو
-
ਪੰਜਾਬੀ
-
Runasimi
-
Gagana Samoa
-
संस्कृत
-
Gaelo Albannach
-
Sepeti
-
Sesotho
-
chiShona
-
سنڌي
-
Soomaali
-
Basa Sunda
-
Wikang Tagalog
-
Тоҷикӣ
-
Татарча
-
తెలుగు
-
ትግንያውያን
-
Xitsonga
-
Türkmençe
-
संस्कृत
-
ئۇيغۇرچە
-
Cymraeg
-
isiXhosa
-
ייִדיש
-
Yorùbá
-
isiZulu
- Thil siam chhuah te
- Trauma System a ni
- Spine System (spine System) a ni
- Damdawi lam Power Tool
- Orthopaedic leh Sports Damdawi lam a ni
- CMF/Maxillofacial System hmanga siam a ni
- Joints System a ni
- Cement nei lo Hemi-Hip Joint System hmanga siam a ni
- Hemi-Hip Joint System hi cement hmanga siam a ni
- Cement tel lo Total Hip Joint System hmanga siam a ni
- Cement hmanga siam Total Hip Joint System a ni
- Cement nei lo Revision Hip System a ni
- Cement hmanga siam thar hip System a ni
- Primary Total Knee System a ni
- Revision Khup zawng zawng System
- Hinge Total Khup System a ni
- Pawn lam atanga Fixation Device hmanga siam
- Sterilization Contain hmanga siam a ni
- A chinfel dan tur
- Trauma System a ni
- Intramedullary Nail hmanga siam a ni
- Locking Plate a awm bawk
- No-Locking Plate a awm bawk
- Spine System (spine System) a ni
- CMF/Maxillofacial System hmanga siam a ni
- Khup leh Hip Joints System a ni
- Pawn lam atanga Fixation System hmanga siam
- Damdawi lam Power Tool
- Orthopaedic leh Sports Damdawi lam a ni
- Sterilization Contain hmanga siam a ni
- Kan Factory a ni
- XC Medico chungchang
- Rawngbawlna te
- Hriatthiamna (Insights) te
- Catalogue hrang hrang a awm