![A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers Gafe min bɛ se ka kɛ ka kolotugudaw ni minɛnw dilanbagaw sɛgɛsɛgɛ]()
Ka fɛn dilanbaga kura sugandi kolotugudaw walima minɛnw kama, o tɛ i n’a fɔ ka biro feerelaw caman cili. Sannifeere se sɔrɔli bɛ se ka wuli ka kɛ sariya labatoli kunkolodimi ye, ka kɛ kɔdimi ye, walima dannaya min bɛ i ka dɔgɔtɔrɔso jatew la.
Nin gafe in jɔra diɲɛ kɔnɔ jatebɔlaw ni dɔgɔtɔrɔso sanni jɛkuluw kama minnu b’a fɛ ka hakilijagabɔ-yɔrɔ dɔ sigi senkan walasa ka kolotugudaw dilanbagaw ka setigiya sɔrɔ — kɛrɛnkɛrɛnnenya la n’i bɛ feere kɛ mara caman kɔnɔ walima n’i bɛ ka dɔ fara i ka baara kan Ameriki Latin (LATAM ) A bɛ sinsin fɛn kan i ka kan ka min ɲini, i bɛ se k’o sɛgɛsɛgɛ cogo min na, ani fɛn minnu ka kan k’i bila ka jɔ dɔɔnin.
Nin barokun in bɛ kuma hadamadenw ka kolotugudaw ni opereli minɛnw kan (a tɛ baganw walima sokɔnɔbaganw ka kolotugudaw furakɛli ye).
Fɛn minnu bɛ ta ka taa ni u ye, olu kunbabaw
Aw bɛ a daminɛ ni sɛbɛnw ye, aw kana layidu ta: jogoɲumanya sigida bonya, minɛnw sariya sira daliluw, ani tugu-tugu ɲɔgɔn kɔ.
FDA 510(k) tɛ diɲɛ bɛɛ ta ye— Class I ni Class II minɛn fanba bɛ se ka bɔ , o la i ka baara ye ka minɛnw danfara ni danfara dantigɛliw sɛgɛsɛgɛ, i kana sɔn 'danfara' ma i n’a fɔ jaabi dafalen.
Aw bɛ ISO 13485 minɛ i n’a fɔ taamasiɲɛ koloma; FDA ka san 2026 jogoɲumanya siratigɛ jiginni bɛ bɛn kosɛbɛ ISO 13485 jigiyaw ma.
Aw bɛ taabolo fila kɛ: (1) ka feerekɛla setigiya ka kɔn misaliw ɲɛ, o kɔfɛ (2) ka kɔrɔsiliw tiɲɛ sani PO fɔlɔ ka kɛ.
Fɛn min bɛ taa ni a ye : Faratiw dɔgɔyali fɛɛrɛ teliyalen ye ka i bɛ fɛn min ɲini feerekɛla bɛɛ fɛ, i ka o kɛ cogo kelen na—o kɔfɛ, i bɛ jaabiw suma ɲɔgɔn na kɛrɛfɛ.
'kolodilabaarakɛlaw' bɛ min ɲɛfɔ tiɲɛ na (ani mun na a nafa ka bon) .
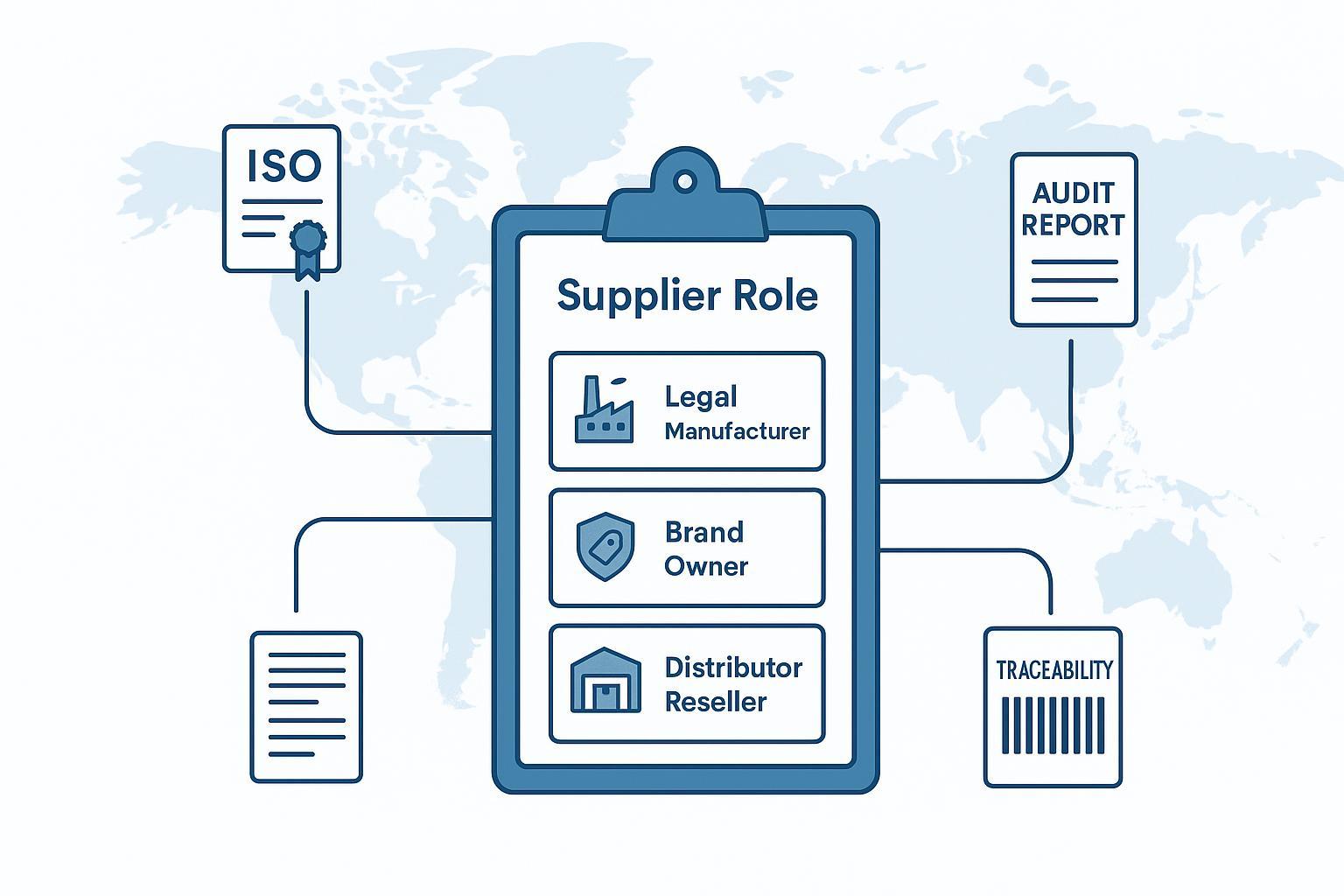
![Ja min bɛ kolotugudaw feerelaw jɔyɔrɔw jira (sariya siratigɛ la, fɛn dilannikɛla, markatigi, feerekɛla) ani sɛbɛnw-fɔlɔ sɛgɛsɛgɛli taabolo]()
Mɔgɔw bɛ 'furakɛla' kɛ ka kɔrɔ wɛrɛw kɛ: fɛn dilannikɛla, bɛnkansɛbɛn dilannikɛla, jagokɛla, walima hali jagokɛla. O daɲɛ fila-fila in ye farati ye.
Ni aw bɛ ka lisi surun dɔ jɔ orthopaedic suppliers , ɲininkali fɔlɔ ka nɔgɔn: Yala aw bɛ ka sariya dilabaga, a taamasiyɛn tigi, walima a feerela/feerekɛla dɔ jateminɛ wa? I ka sɛbɛnw sɛgɛsɛgɛli sɛbɛn bɛ Changé ka kɛɲɛ ni jaabi ye.
Nin fana ye yɔrɔ ye, ekipu caman b’u ka sɔrɔyɔrɔw tila yɔrɔ min na: ɲininkali dɔw bɛ kɛ kolotugudaw dilanbagaw kan (minɛn dafalenw), dɔw bɛ tali kɛ OEM/ODM jɛɲɔgɔnw kan minnu bɛ aw ka sirako bolodalen dɛmɛ. Vetting logic bɛ ɲɔgɔn dafa—nka sɛbɛnw ni kunkanbaaraw tɛ kelen ye.
Sariya waleyali : ni feerekɛla dɔ tɛ se k’u jɔyɔrɔ ɲɛfɔ ka jɛya (a dilannikɛla ni feerekɛla yamaruyalen ni OEM/ODM jɛɲɔgɔn), i ka i ka baara sumaya ani ka jɛya sɔrɔ sani i ka kuma sɔngɔko kan.
Taabolo fɔlɔ : Aw ka kan ka min ɲini sani aw yɛrɛ ka misali ɲini
Nin dakun in ye k’a jira ko feerekɛla ye mɔgɔ sɛbɛ ye, a bɛ se kosɛbɛ, wa a labɛnnen don sɛbɛnw kama—sanni i ka waati jeni misali logistiki kan.
1) Jogoɲumanya siratigɛ daliluw (ani a bonya) .
Aw ye ɲininkali kɛ:
N’aw bɛ ka sɔngɔko caman suma ɲɔgɔn na, aw k’a kɔlɔsi k’a jɛya ni feerekɛla dɔ ye ISO 13485 koloci dilanbaga ye (ani ni seereyaw hakɛ bɛ bɛn aw bɛ fɛn dilannenw denbaya tigitigi minnu sɔrɔ).
Kɔsa in na, jatebɔ cogoya (jɛkulu jumɛn ye seere di, ani a bɛ ban waati min na) .
Jogoɲumanya lajɛba min bɛ sanfɛ : ɲininkaliw ɲɛnabɔli, sariya labatoli kɔlɔsili, waleyaw ɲɛnabɔli
Mun na o nafa ka bon: ISO 13485 ye diɲɛ taamasiyɛn jɛlen dɔ ye min b’a jira ko fɛn dilannikɛla dɔ bɛ jogoɲumanya sira sɛbɛnnen dɔ baara. Dancɛ tigɛ porogaramuw kama (LATAM fana sen bɛ o la), i mago bɛna kɛ dalilu kɛrɛnkɛrɛnnenw na hali bi—o la, i ka ISO 13485 minɛ i n’a fɔ a daminɛcogo, o kɔfɛ, i ka jamana laɲinitaw bɛ min ɲini i tɔgɔ sɛbɛnni, jateminɛw, ani sariya labatoli taabolo la, i k’o sɛgɛsɛgɛ.
2) Regulatory pathway proof ka ɲɛsin fɛn kɛrɛnkɛrɛnnenw ma i bɛ minnu sɔrɔ
'Yala nin fɛn ninnu saniyalen don/u tɔgɔ sɛbɛnna wa?' ye ɲininkali ye min tɛ jɛya. Fisa:
I b’a fɛ ka fɛn dilannenw denbaya kelen-kelen bɛɛ san, i k’a ɲininka minɛn suguya jumɛn bɛ tali kɛ i ka sugu laɲinitaw la ani o bɛ mun jira sariya sira la.
I bɛ feere kɛ jamana minnu na, ɲini sugu kɛrɛnkɛrɛnnenw i ka dalilu kɛrɛnkɛrɛnnenw na (misali la, i tɔgɔ sɛbɛnni/ladɔnniya dalilu, lisi sɛbɛnw, mɔgɔ yamaruyalen/sɛbɛn tigi ka kunnafoniw, walima sɛbɛn wɛrɛw minnu bɛ kɛ fanga fɛ—ka kɛɲɛ ni fanga ye).
Aw bɛ layini kelen fara aw ka dumunifɛnw tacogo sɛbɛn kan min bɛ wele ko sariya sira sɛgɛsɛgɛli ani ka ɲini (a) o fɛn dilannenw denbaya tɔgɔsɛbɛnni/sɛbɛnnisɛbɛn dɔ fɛ aw ka jamana laɲinitaw la, walima (b) basigi sɛbɛnnen dɔ kan min bɛ fɔ 'danfara,' fɛn o fɛn na, dancɛw/dantigɛliw sen bɛ o la.
⚠️ Ladilikan : 'Exempt' kɔrɔ tɛ abada 'unregulated.' A ka c'a la, o kɔrɔ ye ko fɛɛrɛ kɛrɛnkɛrɛnnen dɔ ka kan ka kɛ ka kɔn sugu ɲɛ o minɛn suguya in na o mara kɔnɔ—kɔlɔsili wɛrɛw bɛ baara la hali bi.
3) Traceability ani labeling labɛnni
Aw ye ɲɛfɔli jɛlen ɲini:
Lot/serial traceability (u bɛ tugu cogo min na ka bɔ minɛn dafalen na ka segin fɛnɲɛnamafagalanw ni taabolo la)
Labeling controls (label changements bɛ sɔn cogo min na ani ka bɔ cogo min na) .
UDI labɛnni yɔrɔ min na a bɛ se ka kɛ (danfara kɛrɛnkɛrɛnnenw bɛ ɲɛnabɔ cogo min na ani u bɛ sɛgɛsɛgɛ cogo min na) .
Ni aw bɛ tilatila dɔgɔtɔrɔso sigida caman na, aw bɛ UDI traceability furakɛli minɛnw minɛ i n’a fɔ seko min tɛ se ka kumaɲɔgɔnya kɛ: aw mago bɛ feerekɛla dɔ la min bɛ se ka traceability, foro walew, ani sɛbɛnw ɲinini dɛmɛ k’a sɔrɔ a ma a yɛrɛ labɛn.
Hali n’i mago tɛ labeli pake dafalen na nin waati in na, i b’a fɛ k’a dɔn ko feerekɛla bɛ ni sistɛmu kɔrɔsilen ye—a tɛ miniti laban jatebɔsɛbɛn ye.
4) Fɛnw ni fɛn dilanni kɔlɔsili (supplier-ready version) .
Lafaamuyali waati la, aw mago tɛ fɛɛrɛko dosiye dafalen na. I b’a fɛ ka jaabiw sɔrɔ minnu bɛ kɔrɔsi nin ninnu na:
Fɛn minnu bɛ kɛ ka fɛnw don a kɔnɔ, olu ye jumɛnw ye (misali la, titaniyɔmu sɔgɔlenw, nɛgɛ min tɛ sɔgɔ, PEEK) ani seereyaw bɛ mara cogo di?
Fɛn dilanni taabolo kɔrɔlenw ye jumɛnw ye (masinw dilanni, finiw dilanni, saniya) ani u bɛ sɛgɛsɛgɛ cogo di?
Kɔrɔbɔli/sɛgɛsɛgɛli rapɔɔri jumɛnw bɛ se ka di kulu kelen-kelen bɛɛ la walima komandi kelen-kelen bɛɛ la?
Ni feerekɛla tɛ se ka sɛgɛsɛgɛlikɛyɔrɔw ɲɛfɔ walima ni a tɛ se ka misali rapɔɔri si tila ɲɔgɔn na, o ye taamasyɛn ye.
5) 'min bɛ mun kɛ' karti dɔ OEM/ODM porozɛw kama
N’i bɛ ka private-label, light customization, walima co-development labɛn, i ka ɲininkali kɛ:
Jɔn de ye design control ye?
Jɔn de ye labelling tigi ye?
Jɔn de ye sariyasunba cikanw ni sugu kɔfɛ kɔlɔsili tigi ye?
Ni aw mago bɛ sannifeere hukumu kɔnɔ min ka dun OEM/ODM jateminɛ kama, XC Medico bɛ ni nafolomafɛn caman ye: Gafe laban min bɛ ɲɛsin kolotugudaw OEM & ODM sanni ma.
Taabolo 2nan: I ka kan ka fɛn minnu tiɲɛ sani i ka sanni fɔlɔ kɛ
![What to validate before your first purchase order Fɛn min ka kan ka tiɲɛ sani i ka sanni fɔlɔ kɛ]()
Etape 1 b’a fɔ aw ye ko feerekɛla ka kan ka kɔrɔbɔ. Taabolo 2nan ye ka a lajɛ ko i bɛ se ka sɛgɛsɛgɛli kɛ k’a sɔrɔ kabako ma kɛ.
1) Yɛlɛma kunbɛncogo: i b’a dɔn cogo min na ni fɛn dɔ Changé
Aw ye ɲininkali kɛ feerekɛla bɛ baara kɛ cogo min na:
Ja/spec bɛ Changé (Yɛlɛma).
Fɛnɲɛnɛma feerela bɛ fɛn caman Changer
Taabolo caman cili (misali la, dugukolo sanfɛla furakɛli, saniya) .
O kɔfɛ, aw bɛ ɲininkali nafama kɛ: 'An bɛna ladɔnniya cogo di, ani sɛbɛn jumɛnw bɛ sɔrɔ an fɛ?'
2) Jalakiw ɲɛnabɔli, CAPA, ani foro wale labɛnni
Aw mago t’u ka CAPA dosiye dafalen na. Aw mago bɛ ninnu na:
U ka ɲininkaliw tali taabolo (u bɛ kunnafoni minnu minɛ) .
U bɛ bɛnbaliyakow sɛgɛsɛgɛ cogo min na
U bɛ foroko ko minnu bɛ se ka kɛ, olu lase jɛɲɔgɔnw ma cogo min na
Fɛn dilanbagaw minnu tɛ se ka kololi kɛcogo ɲɛfɔ ɲininkaliw ni latilenni walew kama, a ka gɛlɛn ka da olu la sɛgɛsɛgɛli la.
3) Foroko/sterilize jigiya—ni o bɛ bɛn i ka sanni ma dɔrɔn
Fɛn bɛɛ tɛ feere sterili. Nka ni aw bɛ fɛnw sɔrɔ minnu pake sterili (walima ni aw bɛ aw jigi da feerekɛla kan walasa ka barika sterili dafalen sɔrɔ), aw bɛ:
Ni sterili validation tɛ aw ka seko kɔnɔ, aw bɛ nin yɔrɔ in to a ka nɔgɔn ani ka aw ka baara kɛ ni min ye ni feerekɛla ka baara ye, aw bɛ o sɛbɛn.
4) Logistiki ni Incoterms minnu bɛ bɛn i ka farati muɲuni ma
Aw ye ɲininkali kɛ:
Standard lead times ka kɛɲɛ ni fɛn dilannenw denbaya ye
Backorder kumaɲɔgɔnya jigiyaw
Shipping terms (Incoterms) ani import support (dumuni sɛbɛnw, DDP suganditaw ni u dira)
Nin yɔrɔ in de la 'sɔngɔba kelen-kelen' bɛ dɛsɛ tuma caman na—bari musaka lakika bɛ jira waati latɛmɛni na, cikan yɔrɔ dɔw la, ani kumaɲɔgɔnya min bɛ kɛ ni wale ye.
5) Portfolio fit: yala u bɛ se k’aw ka dɔgɔtɔrɔso kiliyanw dɛmɛ waati jan kɔnɔ wa?
Portfolio caman sɔrɔli bɛ se ka dɔ bɔ feerekɛlaw ka caya la (ani u tɔgɔ sɛbɛnni cɛsiri) waati kɔnɔ.
N’i b’a fɛ ka teliya ka sanity-check product breadth, i bɛ se ka feerekɛla ka category coverage suma ni i bɛ min feere bi. Walasa ka XC Medico ka katalogu-nivo lajɛ, aw ye Products ɲɛ lajɛ.
Darapo bilenman minnu ka kan ka i sumaya
Darapo bilenman bɛɛ tɛ mɔgɔ bɔ mɔgɔ la, nka u kelen-kelen bɛɛ ka kan ka sɛgɛsɛgɛli juguba lawuli.
Sɛbɛn minnu tɛ ni u bonya ye, u dafa don, walima u dicogo jɛkulu
'FDA sɔnna' kumasenw fɔcogo min ma jɛya, min jɛlen don tiɲɛ na, min bɔra, walima min bɛ se ka kɛ
Traceability jaabiw ('an bɛ se k'a nɔfɛ') k'a sɔrɔ u ma cogo ɲɛfɔ
Yɛlɛma kɔlɔsili jigiya sɛbɛnnen si tɛ yen
Layidu tɛmɛnen don lead time kan ni inventory/production plan jɛlen tɛ
Sariya jɔyɔrɔ min ma jɛya (dilanbaga ni feerekɛla) .
XC Medico bɛ bɛn yɔrɔ min na (misali la, feerekɛla ka kunnafoni) .
N’aw b’a fɛ ka misali sɔrɔ, min b’a jira ko feerekɛla dɔ bɛ se k’a yɛrɛ jira cogo min na ka kɛɲɛ ni sɛgɛsɛgɛli sɛbɛn ye min bɛ sanfɛ, XC Medico bɛ jɔyɔrɔ ta i n’a fɔ kolotugudaw ni minɛnw dilannikɛla min ka baarakɛminɛn caman bɛ a bolo (kɔkolo, joginda, kolotugudaw, farikoloɲɛnajɛ fura, kɛnɛma fiɲɛbɔlanw, CMF) ani minɛnw.
U bɛ baarakɛminɛnw ni dɛmɛ fana jira minnu nafa ka bon feerelikɛlaw bolo—sɛbɛnw dɛmɛni, sɛbɛnnikɛminɛnw dɛmɛni suguw la minnu tɛ kelen ye, ani OEM/ODM suganditaw. Walasa ka o siraba lajɛ, aw ye sɛbɛn ɲɛ XC Medico ka OEM/ODM baarakɛlaw.
Nin dakun in tɛ ladilikan ye; o ye misali ye i ka kan ka kuma suguya minnu baara ka kɛ sɛgɛsɛgɛli ɲininkaliw ye.
Fɛn nataw minnu bɛ kɛ
N’i bɛ ka lisi surun dɔ jɔ, fɛɛrɛ min nafa ka bon kosɛbɛ, o ye k’i ka feerekɛla-sɛbɛnni pake kɛ cogo la min bɛ bɛn sariya ma, ka ɲinini kelen ci kandida bɛɛ ma.
Daminɛ-minɛn nɔgɔman dɔ:
Stage 1 doc list (certs, scope, jɔyɔrɔ jɛlen) .
Stage 2 controls list (Yɛlɛma kɔlɔsili, ɲininkaliw/CAPA, logistiki) .
Ɲɛ kelen sumalen jatebɔsɛbɛn min bɛ kɛ ka fɛnw dilanbagaw jate tuma bɛɛ
Furakɛcogo/sariyasenw ka dantigɛli: Nin barokun ye sannifeere kalanni bɛɛ ye, wa a tɛ furakɛli walima sariya ladilikan ye. Fɛn minnu ka kan ka kɛ, olu tɛ kelen ye ka kɛɲɛ ni fɛn dilannen suguya ye ani u ka fanga ye—aw bɛ a sɛgɛsɛgɛ ni sariyasunba/jogoɲumanya baarakɛlaw ye minnu bɛ se kosɛbɛ.
Ɲininkaliw
Yala kolotugudaw bɛɛ bɛ sariyaw ka sɔnni kelen de wajibiya yɔrɔ bɛɛ wa?
Ayi, sariya siraw bɛ danfara ka kɛɲɛ ni jamana ni minɛn suguya ye. Setigiya ka kan ka sinsin sugu kɛrɛnkɛrɛnnen sɛbɛnw kan i bɛ feere yɔrɔ min na (ani baara bɛna kɛ ni fɛn dilannen ye yɔrɔ min na), a man kan ka sinsin fɛn kelen kan min bɛ bɔ fɛn dilanbaga si fɛ.
Yala ISO 13485 bɛ se ka kɛ sababu ye ka feerekɛla dɔ ka se sɔrɔ wa?
O ye daminɛ taamasiyɛn barikama ye, nka a tɛ maana bɛɛ ye. Aw ka kan ka jateminɛ kɛ hali bi ka tugu ɲɔgɔn kɔ, ka fɛn caman Changements control, complaint/CAPA discipline, ani (ni a bɛnnen don) pakew/sterilisation validation ani logistics dannaya.
Filiba jumɛn de bɛ kɛ tilabagaw fɛ ni u bɛ fɛnw feerelaw sɛgɛsɛgɛ?
A bɛ daminɛ ni sɔngɔ ye. Aw bɛ a daminɛ ni jɔyɔrɔ jɛlen ye ani sɛbɛnw labɛnni ye fɔlɔ, o kɔfɛ, aw bɛ misaliw kɔrɔbɔ, o kɔfɛ, aw bɛ sariyaw sigi senkan ni aw ye dɔ bɔ sariya labatoli ni sɔrɔko farati la.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu