![A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers Akwankyerɛ a mfaso wɔ so a wɔde bɛhwehwɛ wɔn a wɔde nnompe a wɔde hyɛ nipadua mu ne nnwinnade a wɔde ma mu]()
Sɛ́ wobɛpaw obi foforo a ɔde nnompe a wɔde hyɛ nipadua mu anaa nnwinnade bɛma no nte sɛ nea wobɛsakra wɔn a wɔtɔn wɔ adwumayɛbea hɔ. Bo a wodi nkonim betumi adan tiyɛ a ɛma wodi mmara so, backorder spiral, anaasɛ ahotoso a ɛbɔ w’ayaresabea akontaabu ahorow.
Wɔakyekye akwankyerɛ yi ama wiase nyinaa akyekyɛfo ne ayaresabea adetɔ akuw a wɔpɛ sɛ wonya nhumu-gyinabea nhyehyɛe a ɛfa nnompe ho aduruyɛfo a wɔfata ho —titiriw sɛ wotɔn kɔ mmeae pii anaasɛ woretrɛw mu wɔ Latin Amerika (LATAM) . Ɛtwe adwene si nea ɛsɛ sɛ wobisa, sɛnea wobɛhwɛ sɛ ɛyɛ nokware, ne nea ɛsɛ sɛ ɛma wugyina kakra so.
Saa asɛm yi fa nnipa nnompe a wɔde hyɛ nipadua mu ne nnwinnade a wɔde yɛ oprehyɛn (ɛnyɛ mmoa anaa afieboa nnompe ho aduruyɛ) ho.
Nneɛma atitiriw a wɔde kɔ
Fi ase de nkrataa, ɛnyɛ bɔhyɛ ahorow: nhyehyɛe a ɛyɛ papa no kɛse, mfiri mmara kwan so adanse, ne nea wotumi hwehwɛ.
FDA 510(k) isn't universal— most Class I and some Class II devices can be exempt , enti w'adwuma ne sɛ wobɛhwɛ mfiri no nkyekyɛmu ne anohyetoɔ a wɔde yi afiri mu no mu, ɛnyɛ sɛ wobɛgye 'exempt' atom sɛ mmuaeɛ a ɛyɛ blanket.
Fa ISO 13485 di dwuma sɛ sɛnkyerɛnne titiriw; FDA 2026 quality-system nsakrae no ne ISO 13485 akwanhwɛ hyia kɛse.
Fa akwan abien di dwuma: (1) fata nea ɔde nneɛma ma no ansa na wɔayɛ nhwɛsode ahorow, afei (2) ma nea wɔde di dwuma no yɛ nokware ansa na wɔayɛ PO a edi kan.
Key Takeaway : Ɔkwan a ɛyɛ ntɛm sen biara a wobɛfa so atew asiane so ne sɛ wode nea wubisa obiara a ɔde nneɛma ma no bɛyɛ nea ɛfata—afei wode mmuae ahorow no bɛtoto nkyɛnkyɛn.
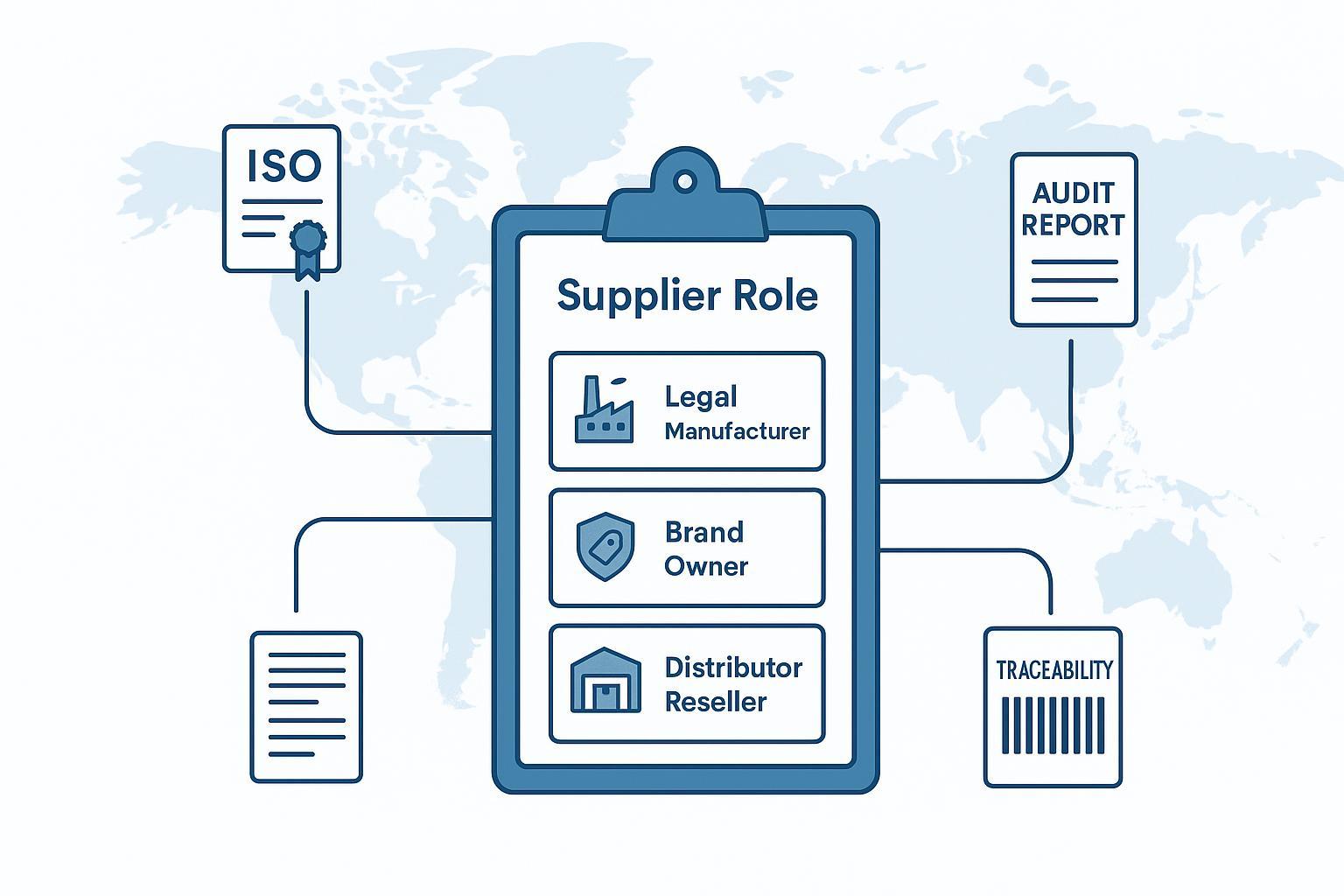
Nea 'orthopaedic suppliers' kata so ankasa (ne nea enti a ɛho hia) .
![Mfonini a ɛkyerɛ nnompe ho aduruyɛfoɔ dwumadie (mmara kwan so yɛfo, ahyɛnsodeɛ wura, nea ɔkyekyɛ) ne nkrataa-di kan vetting flow]()
Nkurɔfoɔ de 'supplier' di dwuma de kyerɛ nneɛma ahodoɔ: adwumfoɔ, apam adwumayɛfoɔ, kyekyɛfoɔ, anaa mpo aguadi adwumakuo. Saa asɛm a emu nna hɔ no yɛ nea asiane wom.
Sɛ worekyekye shortlist a orthopedic suppliers , asɛmmisa a edi kan no yɛ mmerɛw: So woresusuw nea ɔyɛe wɔ mmara kwan so, nea ɔwɔ ahyɛnsode no, anaa nea ɔkyekyɛ/ɔtɔn bio no mu? Wo nkrataa a wode hwehwɛ nneɛma mu no sesa gyina mmuae no so.
Eyi nso ne beae a akuw pii kyekyɛ wɔn sourcing akwan mu: nsɛmmisa bi fa wɔn a wɔde nnompe a wɔde hyɛ nipadua mu (mfiri a wɔawie) ho, bere a afoforo fa OEM/ODM ahokafo a wɔboa wo private-label kwankyerɛ no ho. Vetting logic no di nsɛ —nanso nkrataa ne asɛyɛde ahorow no nyɛ pɛ.
Mmara a mfaso wɔ so: sɛ obi a ɔde nneɛma ma no ntumi nkyerɛkyerɛ wɔn dwumadi mu pefee (ɔyɛfo vs. nea wɔama no tumi sɛ ɔkyekyɛ nneɛma vs. OEM/ODM hokafo), brɛ wo ho ase na nya pefeeyɛ ansa na woaka bo ho asɛm.
Ɔfã 1: Nea ɛsɛ sɛ wobisa ansa na woabisa nhwɛsode mpo
Saa fã yi fa sɛ wobɛkyerɛ sɛ nea ɔde nneɛma ma no yɛ nokware, ɔfata, na wasiesie ne ho sɛ ɔbɛkyerɛw nkrataa —ansa na wobɛhyew bere wɔ nhwɛsode nneɛma a wɔde kɔ baabiara so.
1) Quality system adanse (ne nea ɛkɔ akyiri) .
Bisa sɛ:
Sɛ wode nneɛma pii a wɔde toto ho retoto ho a, hyɛ no nsow pefee sɛ ebia obi a ɔde nneɛma ma no yɛ ISO 13485 nnompe ho adwumayɛfo (ne sɛ ebia adansedi krataa no kɛse ne nneɛma mmusua pɔtee a worehwehwɛ no hyia anaa).
Nnansa yi akontabuo tebea (kuo bɛn na ɛde adansedie krataa no maeɛ, ne berɛ a ɛbɛtwa mu) .
Nhwehwɛmu a ɛkorɔn a ɛfa su pa ho: anwiinwii ho dwumadie, mmara a ɛnhyia sohwɛ, nteɛsoɔ nhyehyɛeɛ
Nea enti a eyi ho hia: ISO 13485 yɛ wiase nyinaa nsɛnkyerɛnne a emu da hɔ sen biara a ɛkyerɛ sɛ nea ɔyɛ no de nhyehyɛe a wɔakyerɛw a ɛfa nneɛma pa ho di dwuma no mu biako. Wɔ nhyehyɛe ahorow a ɛfa ahye so (a LATAM ka ho) ho no, wobɛda so ara hia adanse a ɛfa gua pɔtee bi ho —enti fa ISO 13485 sɛ mfiase, afei hwɛ nea aman a wode w’ani asi so no hwehwɛ ma wɔn dinkyerɛw, akontaabu, ne mmara sodi a ɛkɔ so no mu.
2) Mmara kwan adanse ma nneɛma pɔtee a worehwehwɛ
'So wɔaprapra/wɔakyerɛw saa nneɛma yi?' yɛ asɛmmisa a ɛnyɛ den. Ɛyɛ:
Wɔ nneɛma abusua biara a woayɛ nhyehyɛe sɛ wobɛtɔ fam no, bisa mfiri nkyekyɛmu bɛn na ɛfa wo gua a wode w’ani asi so no so ne nea ɛno kyerɛ ma mmara kwan no.
Hwehwɛ adanse a ɛfa gua pɔtee bi ho ma aman a wotɔn kɔ mu no (sɛ nhwɛso no, adanse a ɛkyerɛ sɛ woakyerɛw wo din/bɔ amanneɛ, adansedi nkrataa a ɛkyerɛ sɛ woakyerɛw wo din, ananmusifo a wɔama no tumi/wɔn a wokura din din ho nsɛm, anaa aban nkrataa afoforo—a egyina tumidi so).
Fa line baako ka wo intake kratasin a wɔfrɛ no regulatory pathway verification mu na hwehwɛ sɛ (a) registration/clearance reference ma saa product abusua no wɔ aman a wode w'ani asi so no mu, anaa (b) documented nnyinaso ma 'exemption,' biara a wɔkyerɛ sɛ ahye/anohyeto ahorow ka ho.
⚠️ Kɔkɔbɔ : 'Exempt' nkyerɛ da sɛ 'unregulated.' Ɛtaa kyerɛ sɛ anammɔn pɔtee bi ansa na wɔatɔn no ho nhia mma saa mfiri no su wɔ saa tumidi no mu—nhyɛso afoforo da so ara di dwuma.
3) Traceability ne labeling ahoboa
Bisa ma wɔmma wo nkyerɛkyerɛmu a emu da hɔ wɔ:
Lot/serial traceability (sɛnea wodi akyi fi mfiri a wɔawie so san kɔ nneɛma a wɔde yɛ adwuma ne akwan horow so)
Labeling controls (sɛnea wɔpene label nsakrae so na woyi no adi) .
UDI ahoboa wɔ baabi a ɛfata (sɛnea wɔhwɛ nkyerɛwde soronko so na wɔdi ho adanse) .
Sɛ wokyekyɛ kɔ ayaresabea nhyehyɛe ahorow pii mu a, fa UDI traceability aduruyɛ mfiri di dwuma sɛ tumi a wontumi nsusuw ho: wuhia obi a ɔde nneɛma ma a obetumi aboa traceability, afuw mu nneyɛe, ne nkrataa a wɔsrɛ a ɔnyɛ nea wɔayɛ no yiye.
Sɛ mpo wunhia label packet a edi mũ wɔ saa bere yi mu a, wopɛ sɛ wuhu sɛ nea ɔde ma no wɔ nhyehyɛe a wɔahyɛ so —ɛnyɛ simma a etwa to spreadsheet.
4) Nneɛma ne nneɛma a wɔyɛ (supplier-ready version) .
Wɔ awareness stage mu no, wunhia mfiridwuma ho dossier a edi mũ. Wopɛ mmuae a wɔahyɛ so ampa wɔ:
Dɛn ne standard implant nneɛma a wɔde di dwuma (sɛ nhwɛso no, titanium alloys, stainless steel, PEEK) na ɔkwan bɛn so na wɔhwɛ certs so?
Dɛn ne akwan a ɛho hia a wɔfa so yɛ nneɛma (mfiri, wie, ahotew) na ɔkwan bɛn so na wɔhwehwɛ mu?
Sɔhwɛ/nhwehwɛmu amanneɛbɔ bɛn na wobetumi de ama wɔ batch biara mu anaasɛ wɔ order biara mu?
Sɛ nea ɔde nneɛma ma no ntumi nkyerɛkyerɛ mmeae a wɔhwɛ nneɛma mu anaasɛ ontumi nkyɛ amanneɛbɔ biara a wɔde yɛ nhwɛso a, ɛno yɛ sɛnkyerɛnne.
5) 'hena na ɔyɛ nea' map ma OEM/ODM nnwuma
Sɛ woreyɛ nhyehyɛe ayɛ private-label, light customization, anaa co-development a, bisa sɛ:
Sɛ wuhia adetɔ nhyehyɛe a emu dɔ ma OEM/ODM nhwehwɛmu a, XC Medico wɔ ade a ɛkɔ akyiri: Akwankyerɛ a etwa to a ɛfa Nnompe OEM & ODM Adetɔ ho.
Ɔfã 2: Nea ɛsɛ sɛ wugye tom ansa na woakra ade a edi kan
![What to validate before your first purchase order Nea ɛsɛ sɛ wogye tom ansa na woakra ade a edi kan]()
Stage 1 ka kyerɛ wo sɛ ɛfata sɛ wɔsɔ nea ɔde nneɛma ma no hwɛ. Stage 2 fa hwɛ a wobɛhwɛ sɛ wobɛtumi ayɛ scale a worennyɛ nwonwa.
1) Change control: sɛnea wuhu bere a biribi sesa
Bisa sɛnea nea ɔde nneɛma ma no di ho dwuma:
Drawing/spec nsakrae ahorow
Nneɛma a wɔde ma no sesa
Nsakrae a ɛba ɔkwan a wɔfa so yɛ adwuma no mu (sɛ nhwɛso no, aduru a wɔde yɛ nneɛma a ɛwɔ soro, ahotew) .
Afei bisa asɛmmisa a mfaso wɔ so: 'Ɔkwan bɛn so na wɔbɛbɔ yɛn amanneɛ, na nkrataa bɛn na yenya?'
2) Anwiinwii ho dwumadie, CAPA, ne afuom adeyɛ ho ahoboa
Wonhia wɔn CAPA fael a edi mũ no. Wohia ampa sɛ:
Wɔn complaint intake flow (data bɛn na wɔkyere) .
Sɛnea wɔhwehwɛ nneɛma a ɛne ne ho nhyia mu
Sɛnea wɔka afuw mu nsɛm a ebetumi aba ho asɛm kyerɛ ahokafo
Ɛyɛ den sɛ wobegye wɔn a wɔde nneɛma ma a wontumi nkyerɛkyerɛ nteɛso loop a wɔde ma anwiinwii ne nteɛso nneyɛe mu no mu ahotoso wɔ nsenia mu.
3) Nneɛma a wɔde kyekyere/sterilization akwanhwɛ—sɛ ɛfata wo adetɔ nkutoo a
Ɛnyɛ ade biara na wɔtɔn a wɔmfa nnuru nni dwuma wom. Nanso sɛ worehwehwɛ nneɛma a wɔde ahyɛ mu a ɛnyɛ sterile (anaasɛ wode wo ho to nea ɔde ma no so sɛ wɔbɛma akwanside a ɛnyɛ sterile no ayɛ nokware) a, bisa sɛ:
Packaging validation nsɛm a wɔaboaboa ano
Ɔkwan a wɔfa so sɔ nneɛma hwɛ wɔ nkyekyɛmu/nneɛma a wɔde fa po so hyɛn mu
Shelf-life adanse kwan a wɔfa so yɛ
Sɛ sterile validation nyɛ wo scope fã a, ma ɔfa yi nyɛ mmerɛw na kyerɛw nea ɛyɛ w’asɛyɛde vs. nea supplier no yɛ asɛyɛde.
4) Logistics ne Incoterms a ɛne wo asiane tolerance hyia
Bisa sɛ:
Standard lead times sɛnea product abusua te
Backorder nkitahodi akwanhwɛ ahorow
Nsɛm a ɛfa nneɛma a wɔde mena ho (Incoterms) ne mmoa a wɔde ba amannɔne (aduan ho nkrataa, DDP akwan horow sɛ wɔde ma a) .
Eyi ne baabi a 'great unit price' taa di nkogu-efisɛ ɛka ankasa no da adi wɔ akyɛde, nneɛma a wɔde mena no fã bi, ne nkitahodi a ɛyɛ adwuma mu.
5) Portfolio fit: wobetumi aboa w’ayaresabea adetɔfo bere tenten?
Portfolio a ɛtrɛw no betumi atew wɔn a wɔde nneɛma ma no trɛw (ne mmɔden a wɔbɔ sɛ wɔbɛkyerɛw wɔn din) so bere a bere kɔ so no.
Sɛ wopɛ sɛ wohwɛ sɛnea nneɛma no trɛw ntɛmntɛm a, wubetumi de nea ɔde nneɛma ma no no fã a wɔakata so no atoto nea wotɔn nnɛ no ho. Sɛ wopɛ XC Medico katalog-level nsusuwii a, hwɛ Products kratafa no.
Frɛko kɔkɔɔ a ɛsɛ sɛ ɛma wo brɛ ase
Ɛnyɛ frankaa kɔkɔɔ biara na ɛma obi mfata, nanso ɛsɛ sɛ emu biara kanyan nokwaredi a emu dɔ.
Adansedi nkrataa a enni scope, da a ɛbɛba awiei, anaa kuw a wɔde ma
'FDA apene so' kasasin a enni pefee wɔ nea wɔaprapra mu ankasa, wɔayi afi mu, anaa ɛfa ho
Vague traceability mmuaeɛ ('yebetumi adi akyi') a ɛnkyerɛkyerɛ sɛnea wɔyɛ no mu
Nsakrae a wɔhwɛ kwan a wɔakyerɛw ato hɔ biara nni hɔ
Bɔhyɛ a ɛboro so wɔ lead time a enni inventory/production nhyehyɛe a emu da hɔ
Mmara kwan so dwuma a emu nna hɔ (ɔyɛfo vs nea ɔtɔn bio) .
Baabi a XC Medico fata (nhwɛso no, nea ɔde nneɛma ma no ho nsɛm) .
Sɛ wopɛ nhwɛsoɔ a ɛkyerɛ sɛdeɛ obi a ɔde nneɛma ma no bɛtumi ada ne ho adi atia nhwehwɛmu a ɛwɔ atifi hɔ no a, XC Medico gyina hɔ sɛ nnompe a wɔde hyɛ nipadua mu ne nnwinnadeɛ yɛfo a ɔwɔ nhyehyɛeɛ ahodoɔ pii (akyi berɛmo, adwenemhaw, nkwaa, agumadiɛ aduru, abɔnten so fixators, CMF) ne nnwinnadeɛ.
Wɔsan nso si nnwuma ne mmoa a ɛho hia ma wɔn a wɔkyekyɛ nneɛma so dua —nkrataa mmoa, nneɛma a wɔde kyerɛw wɔn din mmoa wɔ gua ahorow so, ne OEM/ODM akwan horow. Sɛ wopɛ saa kwan no ho nsɛm nyinaa a, hwɛ XC Medico OEM / ODM nnwuma.
Saa ɔfa yi nyɛ nyansahyɛ; ɛyɛ nhwɛsoɔ a ɛkyerɛ nsɛm ahodoɔ a ɛsɛ sɛ wokyerɛ aseɛ kɔ nsɛmmisa a ɛkyerɛ sɛ ɛyɛ nokware.
Anamɔn a edi hɔ a ɛsɛ sɛ woyɛ
Sɛ worekyekye shortlist a, ade a mfaso wɔ so sen biara ne sɛ wobɛhyehyɛ wo supplier-qualification packet no standardize na wode adesrɛ koro no ara akɔma obiara a ɔpɛ sɛ ɔyɛ ɔkannifo.
Ade a ɛnyɛ den a wɔde fi ase:
Stage 1 doc list (certs, scope, dwumadie a ɛda adi pefee) .
Stage 2 controls list (nsesa control, anwiinwii/CAPA, logistics) .
Kratafa biako ntotoho grid a wɔde ma wɔn a wɔde nneɛma ma no nkontaa bere nyinaa
Aduruyɛ/mmara mu asɛm a wɔka: Saa asɛm yi yɛ adetɔ ho adesua a ɛfa nneɛma a wɔkra ho nyinaa ho na ɛnyɛ aduruyɛ anaa mmara mu afotu. Ɛsono ahwehwɛde ahorow no sɛnea nneɛma no te ne tumidi mu —wo ne mmarahyɛfo/adwumayɛfo a wɔfata a wɔyɛ mmara/ɛyɛ papa hwɛ sɛ ɛyɛ nokware.
FAQ
So nnompe a wɔde hyɛ nipadua mu nyinaa hwehwɛ sɛ mmara kwan so pene koro no ara wɔ baabiara?
Dabi, ɛsono akwan a wɔfa so yɛ mmara no sɛnea ɔman ne mfiri ahorow te. Ɛsɛ sɛ ahwehwɛde no gyina gua pɔtee bi ho nkrataa a ɛkyerɛ baabi a wotɔn (ne baabi a wɔde ade no bedi dwuma) so, na ɛnyɛ sɛ obiara a ɔde nneɛma ma no ka sɛ ɛyɛ nea ɛfata obiara.
So ISO 13485 dɔɔso sɛ ɛbɛma obi a ɔde nneɛma ma no afata?
Ɛyɛ mfiase sɛnkyerɛnne a emu yɛ den, nanso ɛnyɛ asɛm no nyinaa. Ɛda so ara hia sɛ wosɔ traceability, nsakraeɛ sohwɛ, anwiinwii/CAPA nteɛsoɔ, ne (bere a ɛfata) packaging/sterilization validation ne logistics ahotosoɔ.
Dɛn ne mfomso kɛse a wɔn a wɔkyekyɛ nneɛma di bere a wɔrehwɛ wɔn a wɔde nneɛma ma no?
Wofi ase fi bo so. Fi ase fa dwumadie mu pefeeyɛ ne nkrataa ahoboa di kan, afei sɔ nhwɛsoɔ hwɛ, afei di nkitaho wɔ nsɛm ho berɛ a woatew mmara sodi ne nneɛma a wɔde ma ho asiane so.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu