![A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers Mɔfiame Nyui aɖe si Ku Ðe Ƒumeŋuɖui Siwo Wotsɔ Dea Ƒumeŋuɖui Kple Dɔwɔnuwo Dzralawo Ŋu]()
Ame yeye tiatia na ƒumeŋuɖui alo dɔwɔnuwo mele abe ɔfis dzralawo ƒe tɔtrɔ ene o. Asixɔxɔ dzi ɖuɖu ate ŋu atrɔ azu sedziwɔwɔ ƒe taɖuame, megbenyawo ƒe ʋuʋu, alo kakaɖedzi si adze wò kɔdzi ƒe gakɔntawo dzi.
Wotu mɔfiame sia na xexeame katã ƒe mamalawo kple kɔdziƒleha siwo di be woawɔ ɖoɖo ɖe ƒumeŋutinunyalawo ƒe dzedze ŋu le nyanyanana ƒe afɔɖeɖe me—vevietɔ ne èdzraa nu ɖe nuto geɖe me alo nèle kekem ɖe enu le Latin Amerika (LATAM) . Eƒoa nu tso nusi nàbia, alesi nàɖo kpe edzii, kple nusiwo wòle be wòana nàtɔ vie ŋu.
Nyati sia ku ɖe amegbetɔ ƒe ƒumeŋuɖui kple amekoko dɔwɔnuwo ŋu (menye lãwo ƒe ƒumeŋuɖui alo aƒemelãwo ƒe ƒumeŋuɖui o).
Nu vevi siwo woatsɔ ayi teƒe bubuwo
Dze egɔme kple nuŋlɔɖiwo, ke menye ŋugbedodowo o: nyonyome ƒe ɖoɖoa ƒe kekeme, mɔ̃a ƒe ɖoɖowɔwɔ ƒe mɔ ƒe kpeɖodzi, kple alesi woate ŋu akpɔe.
FDA 510(k) isn't universal— most Class I and some Class II devices can be exempt , eyata wò dɔe nye be nàɖo kpe mɔ̃a ƒe hatsotso kple ablɔɖe ƒe seɖoƒewo dzi, ke menye be nàxɔ 'exempt' abe ŋuɖoɖo si wotsɔ blanket ene o.
Bu ISO 13485 abe dzesi vevi aɖe ene; FDA ƒe ƒe 2026 ƒe nyonyome ƒe ɖoɖo ƒe tɔtrɔ sɔ kple ISO 13485 ƒe mɔkpɔkpɔwo wu.
Wɔ afɔɖeɖe eve ƒe ɖoɖo: (1) nàna nudzrala la nadze hafi woawɔ kpɔɖeŋuwo, emegbe (2) nàda asi ɖe mɔxexeɖedɔléle nu dzi hafi woawɔ PO gbãtɔ.
Nu Vevi Siwo Nàtsɔ : Mɔ si dzi nàto aɖe afɔku dzi akpɔtɔ kabakaba wue nye be nàwɔ nusi nèbia tso nudzrala ɖesiaɖe si la ɖe ɖoɖo nu—emegbe nàtsɔ ŋuɖoɖowo asɔ kple wo nɔewo.
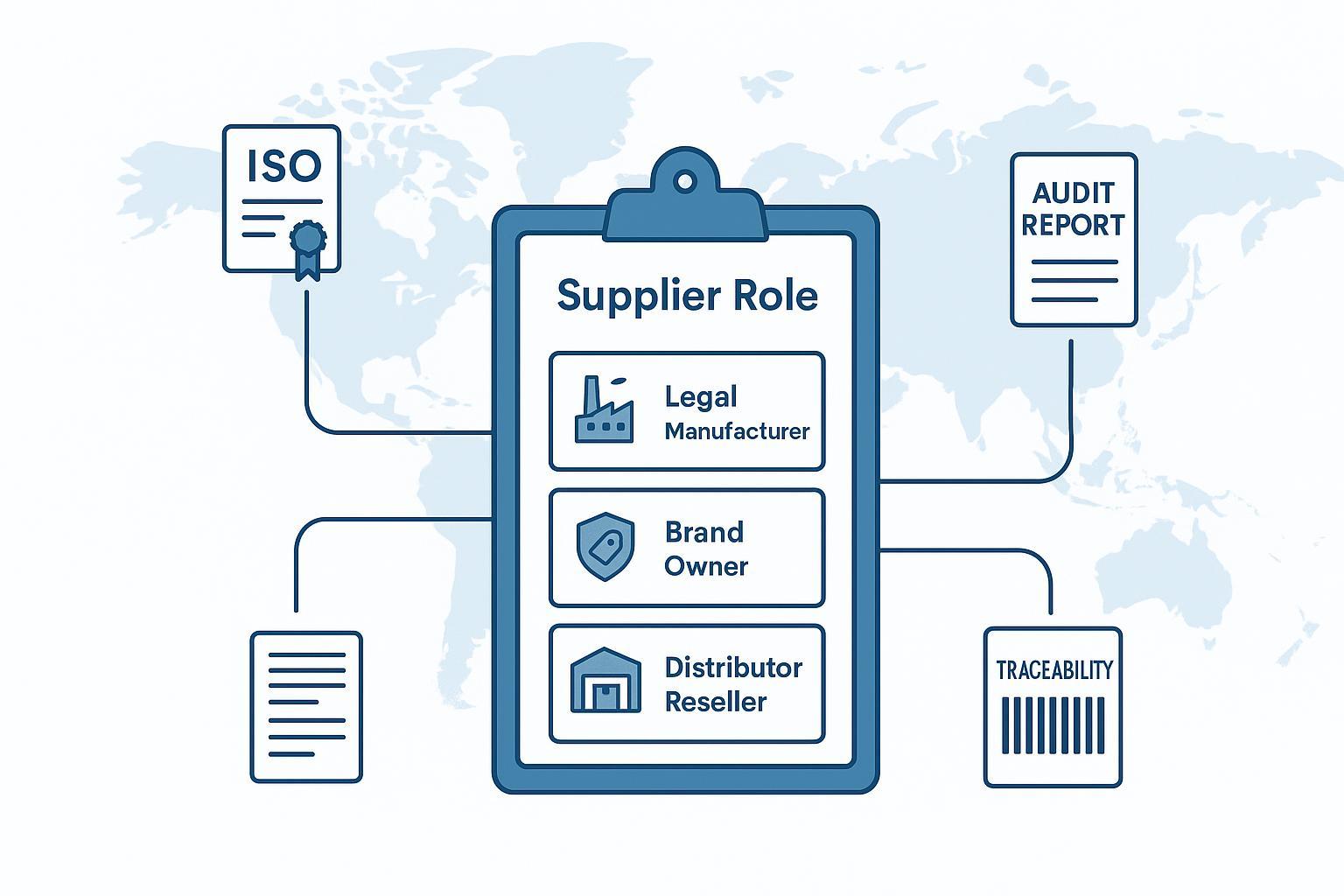
Nusi 'ƒumeŋutinunyalawo' ƒo nu tsoe ŋutɔŋutɔ (kple nusita wòle vevie) .
![Nɔnɔmetata si ɖe ƒumeŋutinunyalawo ƒe akpa siwo wowɔna (sedziwɔla, adzɔnu ƒe ŋkɔ tɔ, mama) kple nuŋlɔɖi-gbãtɔ ƒe vetting flow fia]()
Amewo zãa 'nudzrala' tsɔ fiaa nu vovovowo: adzɔnuwɔla, nubablawɔla, mamala, alo asitsaha gɔ̃ hã. Afɔku le nya ma si me mekɔ o me.
Ne èle xexlẽdzesi kpui aɖe tum la,... orthopaedic suppliers , biabia gbãtɔa le bɔbɔe: Ðe nèle amesi wɔe le se nu, adzɔnuwɔƒea tɔ, alo emama/nudzrala aɖe me dzroma? Wò nuŋlɔɖiwo ƒe ɖaseɖigbalẽa trɔna le ŋuɖoɖoa nu.
Esia hã nye afisi ƒuƒoƒo geɖe ma woƒe dzɔtsoƒemɔwo me le: nyabiase aɖewo ku ɖe ƒumeŋuɖui ƒe atikewo dzralawo (mɔ̃ siwo wowɔ vɔ) ŋu, gake bubuwo ku ɖe OEM/ODM dɔwɔhati siwo doa alɔ wò mɔfiame si nye ame ŋutɔ tɔ ŋu. Vetting logic la sɔ kple wo nɔewo—gake nuŋlɔɖiawo kple agbanɔamedziawo mesɔ o.
Se nyui aɖe: ne nudzrala aɖe mete ŋu ɖe woƒe akpa si wowɔna me nyuie o (nuwɔla kple mamala si ŋu woɖe mɔ ɖo vs. OEM/ODM hadɔwɔla), ɖe edzi nànɔ blewu eye nàna eme nakɔ hafi nàƒo nu tso asixɔxɔ ŋu.
Afɔɖeɖe 1: Nusi nàbia hafi abia kpɔɖeŋuwo gɔ̃ hã
Akpa sia ku ɖe kpeɖodzi nana be nudzrala la nye ame ŋutɔŋutɔ, edze, eye wòle klalo na nuŋlɔɖiwo—hafi nàtɔ dzo ɣeyiɣi ɖe kpɔɖeŋu nudɔdɔwo dzi.
1) Dzɔdzɔmeŋusẽ ƒe ɖoɖo ƒe kpeɖodzi (kple afisi wòkeke ɖo) .
Bia be:
Ne èle asi geɖe tsɔm sɔ kple wo nɔewo la, de dzesii tẽ nenye be nudzrala aɖe nye ISO 13485 ƒumeŋutinunyala (eye nenye be ɖaseɖigbalẽa ƒe lolome sɔ kple adzɔnuwo ƒe ƒome siwo tututu nèle didim tsoe).
Dɔdzikpɔha ƒe nɔnɔme si wowɔ nyitsɔ laa (habɔbɔ si na ɖaseɖigbalẽa, kple ɣeyiɣi si wòwu enu) .
Dzɔdzɔmeŋutete ƒe wɔwɔfia si le ɖoƒe kɔkɔ: nyatoƒoetotowo gbɔ kpɔkpɔ, sedziwɔwɔ mawɔmawɔ dzi kpɔkpɔ, ɖɔɖɔɖo ƒe ɖoɖowo
Nusitae esia le vevie: ISO 13485 nye xexeame katã ƒe dzesi siwo me kɔ wu siwo fia be adzɔnuwɔƒe aɖe zãa nyonyome ƒe ɖoɖo si ŋu woŋlɔ nu tsoe la dometɔ ɖeka. Le ɖoɖo siwo wowɔna le liƒo dzi (siwo dome LATAM hã le) gome la, àgahiã kpeɖodzi siwo ku ɖe asitsanyawo ŋu kokoko—eyata bu ISO 13485 be enye gɔmedzedze, emegbe nàɖo kpe nusi dukɔ siwo ŋu nèɖo taɖodzinu ɖo hiã hena ŋkɔ ŋɔŋlɔ, agbalẽdzikpɔkpɔ, kple sedziwɔwɔ atraɖii dzi.
2) Sedziwɔwɔ mɔ kpeɖodzi na adzɔnu tɔxɛ siwo nèle dzɔtsoƒe
'Ðe woɖe adzɔnu siawo ɖa/woŋlɔ wo ɖe agbalẽ mea?' nye nyabiase si me kɔ. Nyo wu:
Le adzɔnuwo ƒe ƒome ɖesiaɖe si nèɖo be yeaƒle gome la, bia mɔ̃a ƒe hatsotso si sɔ le asi siwo ŋu nèɖo taɖodzinu ɖo me kple nusi ema fia na sedede ƒe mɔa.
Bia asi tɔxɛ ƒe kpeɖodzi na dukɔ siwo me nèdzraa nu le (le kpɔɖeŋu me, ŋkɔ ŋɔŋlɔ/nyatakaka ƒe kpeɖodzi, ŋkɔ ŋɔŋlɔ ƒe ɖaseɖigbalẽwo, ame teƒenɔla/ame si ŋlɔ ŋkɔ ɖe agbalẽ me ƒe nyatakaka siwo ŋu woɖe mɔ ɖo, alo dziɖuɖua ƒe nuŋlɔɖi bubuwo—le ʋɔnudrɔ̃ƒea nu).
Tsɔ fli ɖeka kpe ɖe wò nuɖuɖu ƒe agbalẽvi si woyɔna be ɖoɖowɔɖi ƒe mɔ ƒe kpeɖodzi ŋu eye nàbia be (a) woŋlɔ ŋkɔ/ɖe mɔ na adzɔnuwo ƒe ƒome ma le dukɔ siwo me nèɖo taɖodzinu ɖo me, alo (b) gɔmeɖoanyi si woŋlɔ ɖi na 'woɖe asi le eŋu,' ɖesiaɖe si wogblɔ be liƒowo/seɖoƒewo hã le.
⚠️ Nuxlɔ̃ame : 'Exempt' mefia gbeɖe 'unregulated.' Zi geɖe la, efia be afɔɖeɖe tɔxɛ aɖe do ŋgɔ na asitsatsa mehiã na mɔ̃ ƒomevi ma le ʋɔnudrɔ̃ƒe ma o—dziɖuɖu bubuwo gakpɔtɔ le dɔ wɔm.
3) Traceability kple labeling dzadzraɖo
Bia be woaɖe nu me nyuie tso:
Lot/serial traceability (alesi wokplɔa wo ɖo tso mɔ̃ si wowɔ vɔ dzi yia nu xoxowo kple dɔwɔwɔwo dzi)
Labeling controls (alesi woda asi ɖe label ƒe tɔtrɔwo dzi eye woɖea asi le wo ŋu) .
UDI ƒe dzadzraɖo le afisi wòsɔ le (alesi wokpɔa dzesidenu tɔxɛwo dzi eye woɖoa kpe wo dzii) .
Ne èmae ɖe kɔdzi ƒe ɖoɖo geɖewo me la, bu UDI ƒe atikewɔwɔ ƒe mɔ̃ siwo woate ŋu akpɔ ŋudɔ abe ŋutete si ŋu womate ŋu awɔ ɖoɖo ɖo o ene: èhiã na nudzrala si ate ŋu ado alɔ didi, agbledelawo ƒe afɔɖeɖewo, kple nuŋlɔɖiwo biabia evɔ màwɔ asitɔtrɔ le wo ŋu o.
Ne mèhiã dzesidegbalẽvi blibo le afɔɖeɖe sia me o hã la, àdi be yeanya be ɖoɖo aɖe si dzi wokpɔna le nudzrala la si—menye aɖabaƒoƒo mamlɛtɔ ƒe agbalẽvi o.
4) Nuwo kple wɔwɔme dzi kpɔkpɔ (nudzrala-dzra ɖo ƒe tɔtrɔ) .
Le nyanyanana ƒe afɔɖeɖe me la, mèhiã mɔ̃ɖaŋununya ŋuti nyatakaka blibo o. Èdi be yeakpɔ ŋuɖoɖo siwo dzi wokpɔ ŋusẽ ɖo na:
Nukae nye nusiwo wozãna tsɔ dea ame ƒe ŋutilã me (le kpɔɖeŋu me, titanium alloys, stainless steel, PEEK) eye aleke woléa be na certs?
Nukae nye nuwo wɔwɔ ƒe mɔnu veviwo (mɔ̃wo wɔwɔ, wo wuwu, wo ŋu kɔklɔ) eye aleke woléa ŋku ɖe wo ŋui?
Dodokpɔ/dzidzedzekpɔkpɔ ŋuti nyatakaka kawoe woate ŋu ana le hatsotso ɖesiaɖe me alo le nudɔdɔ ɖesiaɖe me?
Ne nudzrala la mete ŋu ɖɔa ŋkuléleɖenuŋu ƒe dɔwɔƒewo o alo mete ŋu ma kpɔɖeŋunyatakaka aɖeke o la, ke dzesie nye ema.
5) 'amesi wɔa nusi' anyigbatata na OEM/ODM dɔwo
Ne èle ɖoɖo wɔm ɖe private-label, light customization, alo co-development ŋu la, bia be:
Ne èhiã na nuƒle ƒe ɖoɖo si de to wu na OEM/ODM ƒe dodokpɔ la, dɔwɔnu si me kɔ wu le XC Medico si: Mɔfiame Mlɔetɔ na Ƒumeŋutinunya OEM & ODM Nuƒle.
Afɔɖeɖe 2: Nusiwo dzi nàda asi ɖo hafi nàƒle nu gbãtɔ
![What to validate before your first purchase order Nusiwo dzi nàda asi ɖo hafi nàƒle nu gbãtɔ]()
Afɔɖeɖe 1 gblɔ na wò be edze be woado nudzrala la kpɔ. Afɔɖeɖe 2 lia ku ɖe egbɔkpɔkpɔ be yeate ŋu adzidze nu wɔnuku aɖeke manɔmee ŋu.
1) Trɔtrɔ dzi ɖuɖu: alesi nènya ne nane trɔ
Bia alesi nudzrala la wɔa nu ɖe:
Emegbe bia nya si woate ŋu awɔ: 'Aleke woana míanya, eye nuŋlɔɖi kawoe míaxɔ?'
2) Nyatoƒoetotowo gbɔ kpɔkpɔ, CAPA, kple dzadzraɖo ɖe gbeadzi afɔɖeɖe ŋu
Mèhiã woƒe CAPA faɛl bliboa o. Èhiã be:
Woƒe nyatoƒoetoto xɔxlɔ̃ ƒe sisi (nyatakaka siwo woxɔna) .
Alesi wokua nusiwo mewɔ ɖeka o me
Alesi wogblɔa gbeadzinya siwo ate ŋu ado mo ɖa na hadɔwɔlawoe
Esesẽna be woaka ɖe nudzrala siwo mate ŋu aɖɔ tohehe ƒe mɔnu si woatsɔ awɔ nyatoƒoetotowo kple ɖɔɖɔɖodɔwo o la dzi le agbɔsɔsɔ me.
3) Nubablɛ/dɔlékuiwutike ƒe mɔkpɔkpɔwo—ne esɔ ɖe wò nuƒle nu ko
Menye atike ɖesiaɖee wodzrana si me dɔlékui mele o. Gake ne èle nusiwo me dɔlékui mele o dim tsoe (alo èle ŋu ɖom ɖe nudzrala ŋu be wòana mɔxenu si me dɔlékui mele o nanɔ anyi) la, bia be:
Packaging validation ƒe kpukpuiwo
Mama/meliɖoɖo dodokpɔ mɔnu
Nusiwo wodzra ɖo ƒe kpeɖodzi ƒe mɔnu
Ne sterile validation menye wò scope ƒe akpa aɖeke o la, na akpa sia nanɔ bɔbɔe eye nàŋlɔ nusiwo ŋu wò agbanɔamedzi le vs. nusi dzi nudzrala la ƒe agbanɔamedzi le la ɖi.
4) Logistics kple Incoterms siwo sɔ kple wò afɔku dzi ɖuɖu
Bia be:
Standard lead times le adzɔnuwo ƒe ƒomea nu
Backorder kadodo ƒe mɔkpɔkpɔwo
Nusiwo woatsɔ aɖoe ɖe amewo (Incoterms) kple kpekpeɖeŋunana ɖe nusiwo wotsɔ tso duta vɛ ŋu (adzɔxexe ƒe agbalẽwo, DDP ƒe tiatiawɔblɔɖe ne wotsɔe na)
Afi siae 'great unit price' do kpo nu le zi geɖe—elabena gazazã ŋutɔŋutɔ dzena le heheɖemegbe, akpa aɖewo ɖoɖoɖa, kple kadodo si wɔa nu ɖe ame ŋu me.
5) Portfolio fit: ɖe woate ŋu ado alɔ wò kɔdzi asisiwo ɣeyiɣi didia?
Gaxɔmenu siwo keke ta ate ŋu aɖe nudzralawo ƒe kaka (kple agbagbadzedze be woaŋlɔ ŋkɔ) dzi akpɔtɔ le ɣeyiɣi aɖe megbe.
Ne èdi be yealé ŋku ɖe adzɔnuwo ƒe kekeme ŋu kaba la, àte ŋu atsɔ nudzrala la ƒe hatsotso ƒe nugblẽfexeɖoɖo asɔ kple nusiwo nèdzrana egbea. Ne èdi XC Medico ƒe agbalẽdzraɖoƒe ƒe ɖoɖo ƒe wɔwɔfia la, kpɔ Adzɔnuwo ƒe axa.
Aflaga dzĩ siwo wòle be nàɖe wò blewu
Menye aflaga dzĩ ɖesiaɖee ɖee fia be womedze o, gake ele be wo dometɔ ɖesiaɖe nahe kpeɖodzi si de to wu vɛ.
Ðaseɖigbalẽ siwo ƒe lolome, ŋkeke si dzi woawu enu, alo dɔwɔƒe si naa wo mele o
'FDA da asi ɖe' nyagbewo me kɔ nyuie le nusi ŋu woɖe asi le ŋutɔŋutɔ, woɖe asi le eŋu, alo nusi sɔ ŋu o
Vague traceability ŋuɖoɖowo ('míate ŋu akplɔe ɖo') evɔ womeɖɔ alesi woawɔe o
Tɔtrɔ dzi ɖuɖu ƒe mɔkpɔkpɔ siwo woŋlɔ ɖi aɖeke meli o
Ŋugbedodo fũu akpa le ɣeyiɣi si woatsɔ awɔ dɔe ŋu nudzraɖoƒe/nuwɔwɔ ƒe ɖoɖo si me kɔ manɔmee
Akpa si wowɔ le se nu si me mekɔ o (nuwɔla vs nudzrala) .
Afisi XC Medico sɔ le (kpɔɖeŋu si nye nudzrala ƒe nɔnɔmetata) .
Ne èdi be yeakpɔ alesi nudzrala aɖe ate ŋu atsɔ wo ɖokui afia ɖe ɖaseɖigbalẽ si le etame ŋu la, XC Medico ɖo ɖoƒe si nye ƒumeŋuɖui kple dɔwɔnuwo wɔƒe si si ɖoɖo vovovowo ƒe agbalẽdzraɖoƒe le (akɔta, nuveviwɔame, ƒunukpeƒe, kamedefefe ƒe atike, gotagome fixators, CMF) kple dɔwɔnuwo.
Wohea gbe ɖe subɔsubɔdɔwo kple kpekpeɖeŋu siwo le vevie na mamalawo hã dzi—nuŋlɔɖiwo ƒe kpekpeɖeŋu, ŋkɔ ŋɔŋlɔ ƒe nuzazãwo ƒe kpekpeɖeŋu le asi vovovowo me, kple OEM/ODM ƒe tiatiawɔblɔɖewo. Ne èdi mɔ ma ŋuti nyatakaka kpui aɖe la, kpɔ XC Medico ƒe OEM / ODM dɔwɔƒewo.
Akpa sia menye kafukafunya o; enye nya siwo ƒomevi wòle be nàɖe gɔme ɖe kpeɖodzibiabia me ƒe kpɔɖeŋu.
Afɔɖeɖe siwo kplɔe ɖo
Ne èle xexlẽdzesi kpui aɖe tum la, afɔɖeɖe si ŋu viɖe le wue nye be nàwɔ wò nudzrala ƒe dzedze ƒe agbalẽvi la ɖe ɖoɖo nu eye nàɖo biabia ma ke ɖe ame ɖesiaɖe si di be yeaxɔ dɔa.
Gɔmedzedze ƒe dɔwɔnu bɔbɔe aɖe:
Stage 1 doc list (certs, afisi wòakeke ɖo, akpa si woawɔ ƒe eme kɔ) .
Afɔɖeɖe 2 ƒe dziɖuɖuwo ƒe xexlẽdzesi (trɔtrɔ dziɖuɖu, nyatoƒoetotowo/CAPA, ɖoɖowɔwɔ) .
Axa ɖeka ƒe nɔnɔmetata si wotsɔ sɔ kple wo nɔewo hena dzesidede nudzralawo ɣesiaɣi
Atikewɔwɔ/sedede ƒe adzɔgbeɖeɖe: Nyati sia nye nuƒle ŋuti hehenana amewo katã eye menye atikewɔwɔ alo senyawo ŋuti aɖaŋuɖoɖoe wònye o. Nudidiwo toa vovo le adzɔnuwo ƒe ƒomevi kple ʋɔnudrɔ̃ƒe ƒe ŋusẽ nu—ɖo kpe edzi kple sedzikpɔla bibi siwo kpɔa ɖoɖowɔwɔ/nyonyo dzi.
Nyabiasewo ƒe Nyabiasewo
Ðe ƒumeŋuɖuiwo katã hiã be woada asi ɖe edzi le se nu ɖeka ma ke le afisiafia?
Ao, mɔ siwo dzi wotona sea nu la toa vovo le dukɔ kple mɔ̃ ƒomevi aɖe nu. Ele be woatu dzedze ɖe asi tɔxɛ ŋuti nuŋlɔɖi siwo ku ɖe afisi nèdzraa nu le (kple afisi woazã adzɔnua le) dzi, ke menye ɖe nudzrala aɖeke ƒe nya si wògblɔ be esɔ na amewo katã dzi o.
Ðe ISO 13485 sɔ gbɔ be wòadze na nudzrala aɖea?
Enye gɔmedzedze ƒe dzesi sesẽ aɖe, gake menye ŋutinya bliboae o. Ele kokoko be nàda alesi woate ŋu akpɔe, tɔtrɔwo dzi kpɔkpɔ, nyatoƒoetoto/CAPA ƒe amehehe, kple (ne esɔ) babla/dɔlékuiwutikewo ƒe kpeɖodzi kple nudɔdɔwo ƒe kakaɖedzi kpɔ.
Nukae nye vodada gãtɔ si mamalawo wɔna ne wole nudzralawo me dzrom?
Dze egɔme tso asixɔxɔ dzi. Dze egɔme kple akpa si me kɔ kple nuŋlɔɖiwo dzadzraɖo gbã, emegbe nàdo kpɔɖeŋuwo kpɔ, emegbe nàdzro nyawo me ne ènya ɖe sedziwɔwɔ kple nuzazãwo ƒe afɔku dzi kpɔtɔ ko.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu