![A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers]()
Choosing a new supplier for orthopedic implants or instruments isn’t like switching office vendors. A pricing win can turn into a compliance headache, a backorder spiral, or a credibility hit with your hospital accounts.
This guide is built for global distributors and hospital procurement teams who want an awareness-stage framework for orthopedic supplier qualification—especially if you sell into multiple regions or you’re expanding in Latin America (LATAM). It focuses on what to request, how to verify it, and what should make you pause.
This article is about human orthopedic implants and surgical instruments (not veterinary or pet orthopedics).
Key takeaways
Start with documents, not promises: quality system scope, device regulatory pathway evidence, and traceability.
FDA 510(k) isn’t universal—most Class I and some Class II devices can be exempt, so your job is to verify the device classification and the exemption limits, not to accept “exempt” as a blanket answer.
Treat ISO 13485 as a core signal; the FDA’s 2026 quality-system shift aligns more closely to ISO 13485 expectations.
Run a two-stage process: (1) qualify the supplier before samples, then (2) validate controls before the first PO.
Key Takeaway: The fastest way to reduce risk is to standardize what you ask every supplier for—then compare answers side by side.
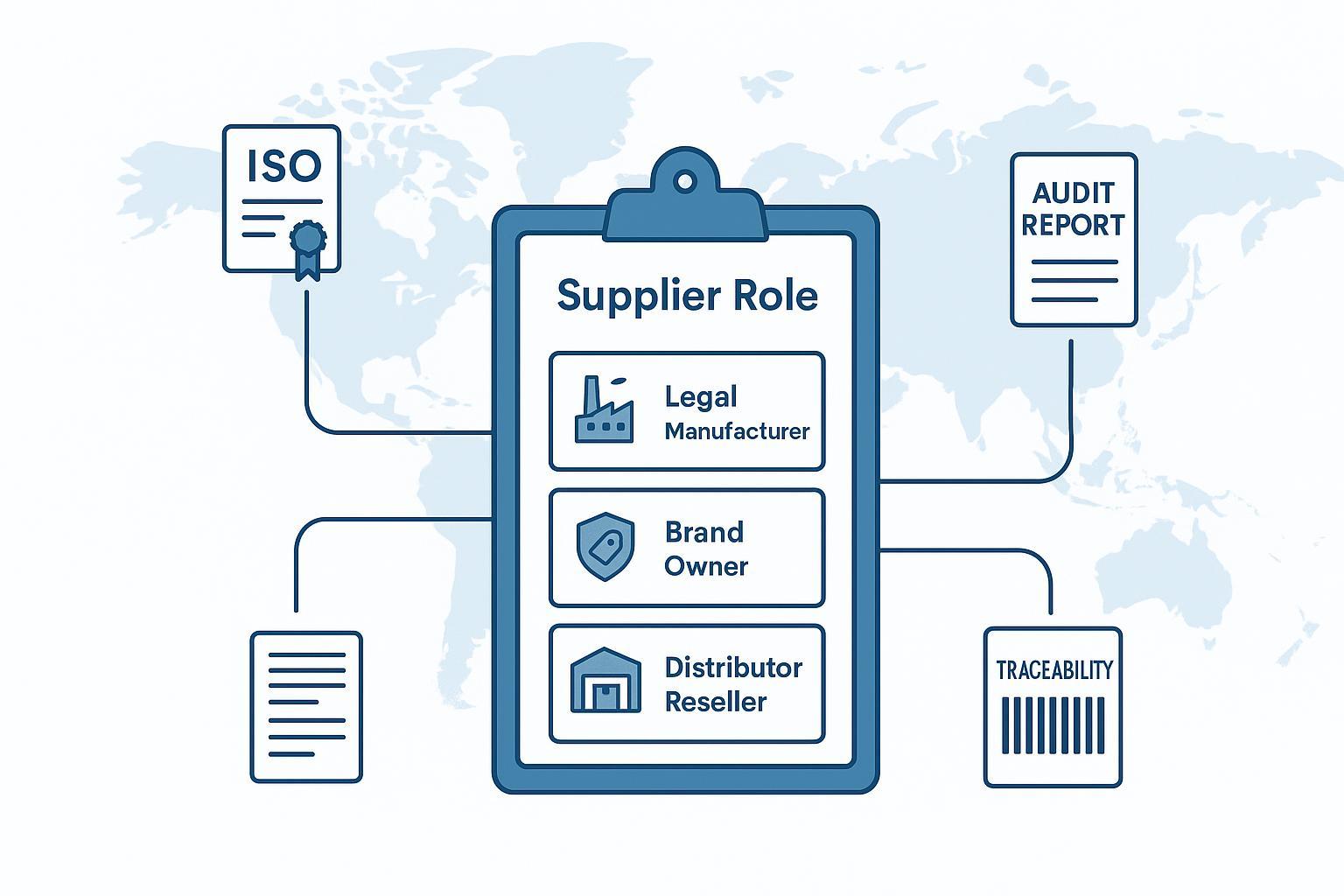
What “orthopedic suppliers” actually covers (and why it matters)
![Illustration showing orthopedic supplier roles (legal manufacturer, brand owner, distributor) and the documentation-first vetting flow]()
People use “supplier” to mean different things: a manufacturer, a contract manufacturer, a distributor, or even a trading company. That ambiguity is risky.
If you’re building a shortlist of orthopedic suppliers, the first question is simple: Are you evaluating the legal manufacturer, the brand owner, or a distributor/reseller? Your documentation checklist changes depending on the answer.
This is also the point where many teams split their sourcing lanes: some queries are about orthopedic implant suppliers (finished devices), while others are about OEM/ODM partners supporting your private-label roadmap. The vetting logic overlaps—but the documents and responsibilities aren’t identical.
A practical rule: if a supplier can’t clearly explain their role (manufacturer vs. authorized distributor vs. OEM/ODM partner), slow down and get clarity before you talk pricing.
Stage 1: What to request before you even ask for samples
This stage is about proving the supplier is real, qualified, and document-ready—before you burn time on sample logistics.
1) Quality system evidence (and scope)
Ask for:
If you’re comparing multiple bids, explicitly note whether a supplier is an ISO 13485 orthopedic supplier (and whether the certificate scope matches the exact product families you’re sourcing).
Recent audit status (what body issued the certificate, and when it expires)
A high-level quality overview: complaint handling, nonconformance controls, corrective action processes
Why this matters: ISO 13485 is one of the clearest global signals that a manufacturer runs a documented quality system. For cross-border programs (including LATAM), you’ll still need market-specific evidence—so treat ISO 13485 as the baseline, then verify what your target countries require for registration, audits, and ongoing compliance.
2) Regulatory pathway proof for the specific products you’re sourcing
“Are these products cleared/registered?” is a blunt question. Better:
For each product family you plan to buy, ask which device classification applies in your target markets and what that implies for the regulatory pathway.
Require market-specific evidence for the countries you sell into (e.g., registration/notification proof, listing certificates, authorized representative/registration holder details, or other official documentation—depending on the jurisdiction).
Add one line in your intake form called regulatory pathway verification and require either (a) a registration/clearance reference for that product family in your target countries, or (b) the documented basis for any claimed “exemption,” including the boundaries/limitations.
⚠️ Warning: “Exempt” never means “unregulated.” It usually means a specific premarket step isn’t required for that device type in that jurisdiction—other controls still apply.
3) Traceability and labeling readiness
Ask for a clear description of:
Lot/serial traceability (how they track from finished device back to raw materials and processes)
Labeling controls (how label changes are approved and released)
UDI readiness where applicable (how unique identifiers are managed and verified)
If you distribute into multiple hospital systems, treat UDI traceability medical devices as a non-negotiable capability: you need a supplier that can support traceability, field actions, and documentation requests without improvising.
Even if you don’t need a full label packet at this stage, you want to know the supplier has a controlled system—not a last-minute spreadsheet.
4) Materials and manufacturing controls (supplier-ready version)
At awareness stage, you don’t need a full technical dossier. You do want controlled answers to:
What are the standard implant materials used (e.g., titanium alloys, stainless steel, PEEK) and how are certs maintained?
What are the critical manufacturing processes (machining, finishing, cleaning) and how are they inspected?
What testing/inspection reports can be provided per batch or per order?
If the supplier can’t describe inspection checkpoints or can’t share any sample reports, that’s a signal.
5) A “who does what” map for OEM/ODM projects
If you’re planning private-label, light customization, or co-development, ask:
If you need a deeper procurement framework for OEM/ODM evaluation, XC Medico has a detailed resource: Ultimate Guide to Orthopedic OEM & ODM Procurement.
Stage 2: What to validate before your first purchase order
![What to validate before your first purchase order What to validate before your first purchase order]()
Stage 1 tells you the supplier is worth testing. Stage 2 is about making sure you can scale without surprises.
1) Change control: how you find out when something changes
Ask how the supplier handles:
Drawing/spec changes
Material supplier changes
Process changes (e.g., surface treatment, cleaning)
Then ask the practical question: “How will we be notified, and what documentation do we receive?”
2) Complaint handling, CAPA, and field action readiness
You don’t need their full CAPA file. You do need:
Their complaint intake flow (what data they capture)
How they investigate nonconformities
How they communicate potential field issues to partners
Suppliers who can’t describe a disciplined loop for complaints and corrective actions are hard to trust at scale.
3) Packaging/sterilization expectations—only if relevant to your purchase
Not every product is sold sterile. But if you are sourcing sterile-packed products (or relying on the supplier for sterile barrier integrity), ask for:
Packaging validation summaries
Distribution/shipping testing approach
Shelf-life evidence approach
If sterile validation isn’t part of your scope, keep this section simple and document what you’re responsible for vs. what the supplier is responsible for.
4) Logistics and Incoterms that match your risk tolerance
Ask for:
Standard lead times by product family
Backorder communication expectations
Shipping terms (Incoterms) and import support (customs paperwork, DDP options if offered)
This is where “great unit price” often fails—because the real cost shows up in delays, partial shipments, and reactive communication.
5) Portfolio fit: can they support your hospital customers long-term?
Broad portfolio coverage can reduce supplier sprawl (and registration effort) over time.
If you want to quickly sanity-check product breadth, you can compare the supplier’s category coverage against what you sell today. For XC Medico’s catalog-level overview, see the Products page.
Red flags that should slow you down
Not every red flag is disqualifying, but each one should trigger deeper verification.
Certificates without scope, expiry date, or issuing body
“FDA approved” phrasing without clarity on what is actually cleared, exempt, or applicable
Vague traceability answers (“we can track it”) without describing how
No documented change control expectations
Overpromising on lead time without a clear inventory/production plan
Unclear legal role (manufacturer vs reseller)
Where XC Medico fits (example supplier profile)
If you want an example of how a supplier might present themselves against the checklist above, XC Medico positions as an orthopedic implants and instruments manufacturer with a multi-system portfolio (spine, trauma, joint, sports medicine, external fixators, CMF) and instruments.
They also highlight services and support that matter to distributors—documentation support, registration material support in different markets, and OEM/ODM options. For an overview of that lane, see XC Medico’s OEM/ODM services.
This section isn’t a recommendation; it’s an example of the kinds of claims you should translate into verification questions.
Next steps
If you’re building a shortlist, the most useful move is to standardize your supplier-qualification packet and send the same request to every candidate.
A simple starter kit:
Stage 1 doc list (certs, scope, role clarity)
Stage 2 controls list (change control, complaints/CAPA, logistics)
A one-page comparison grid for scoring suppliers consistently
Medical/regulatory disclaimer: This article is for general procurement education and is not medical or legal advice. Requirements vary by product type and jurisdiction—verify with qualified regulatory/quality professionals.
FAQ
Do all orthopedic implants require the same regulatory approval everywhere?
No. Regulatory pathways vary by country and device type. Qualification should be based on market-specific documentation for where you sell (and where the product will be used), not on a one-size-fits-all claim from any supplier.
Is ISO 13485 enough to qualify a supplier?
It’s a strong starting signal, but it’s not the whole story. You still need to evaluate traceability, change control, complaint/CAPA discipline, and (when relevant) packaging/sterilization validation and logistics reliability.
What’s the biggest mistake distributors make when vetting suppliers?
Starting with price. Start with role clarity and documentation readiness first, then test samples, then negotiate terms once you’ve reduced compliance and supply risk.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu