![A Practical Guide to Vetting Orthopedic Implant and Instrument Suppliers Wan Praktikal Gayd fɔ Vet Ɔtpidik Implant ɛn Instrumɛnt Splayers]()
Fɔ pik nyu saplay fɔ ɔtpidik implant ɔ inschrumɛnt nɔ tan lɛk fɔ chenj ɔfis vendor. Wan prayz win kin tɔn to kɔmplians ed-ak, bakɔda spiral, ɔ kredibiliti hit wit yu ɔspitul akɔn.
Dis gayd na fɔ di glob ɔl distribyushɔn ɛn ɔspitul prokyumɛnt tim dɛn we want wan ɔwe-stej fɔm fɔ ɔtpidik spɔlayt kwalifayeshɔn—especially if yu de sɛl na bɔku rijyɔn ɔ yu de expand na Latin Amɛrika (LATAM) . I de tɔk mɔ bɔt wetin fɔ aks fɔ, aw fɔ chɛk am, ɛn wetin fɔ mek yu stɔp.
Dis atikul na bɔt mɔtalman ɔtpidik implant ɛn ɔspitul inschrumɛnt (nɔto vetɛriana ɔ pet ɔtpidik).
Ki tek-away dɛn
Start wit dɔkyumɛnt, nɔto prɔmis: kwaliti sistɛm skɔp, divays rigyuletɔri path ɛvidɛns, ɛn traysabiliti.
FDA 510(k) nɔto fɔ ɔlman— bɔku pan di Klas I ɛn sɔm Klas II divays dɛn kin gɛt ɛksɛmpt , so yu wok na fɔ chɛk di divays klasifikeshɔn ɛn di ɛksɛmpshɔn limit, nɔto fɔ aksept 'ɛksɛmpt' as blankit ansa.
Trit ISO 13485 as wan kɔr signal; di FDA in 2026 kwaliti-sistim shift alayns mɔ klos to ISO 13485 ɛkspɛkteshɔn.
Rɔn wan tu-stej prɔses: (1) kwalifay di saplay bifo sampul, dɔn (2) validet kɔntrol bifo di fɔs PO.
Key Takeaway : Di fastest we fɔ ridyus risk na fɔ standad wetin yu aks ɛvri spɔlayt fɔ—dɛn kɔmpia ansa dɛn sayd to sayd.
Wetin 'ɔtpidik spɔlayt dɛm' rili kɔba (ɛn wetin mek i impɔtant)
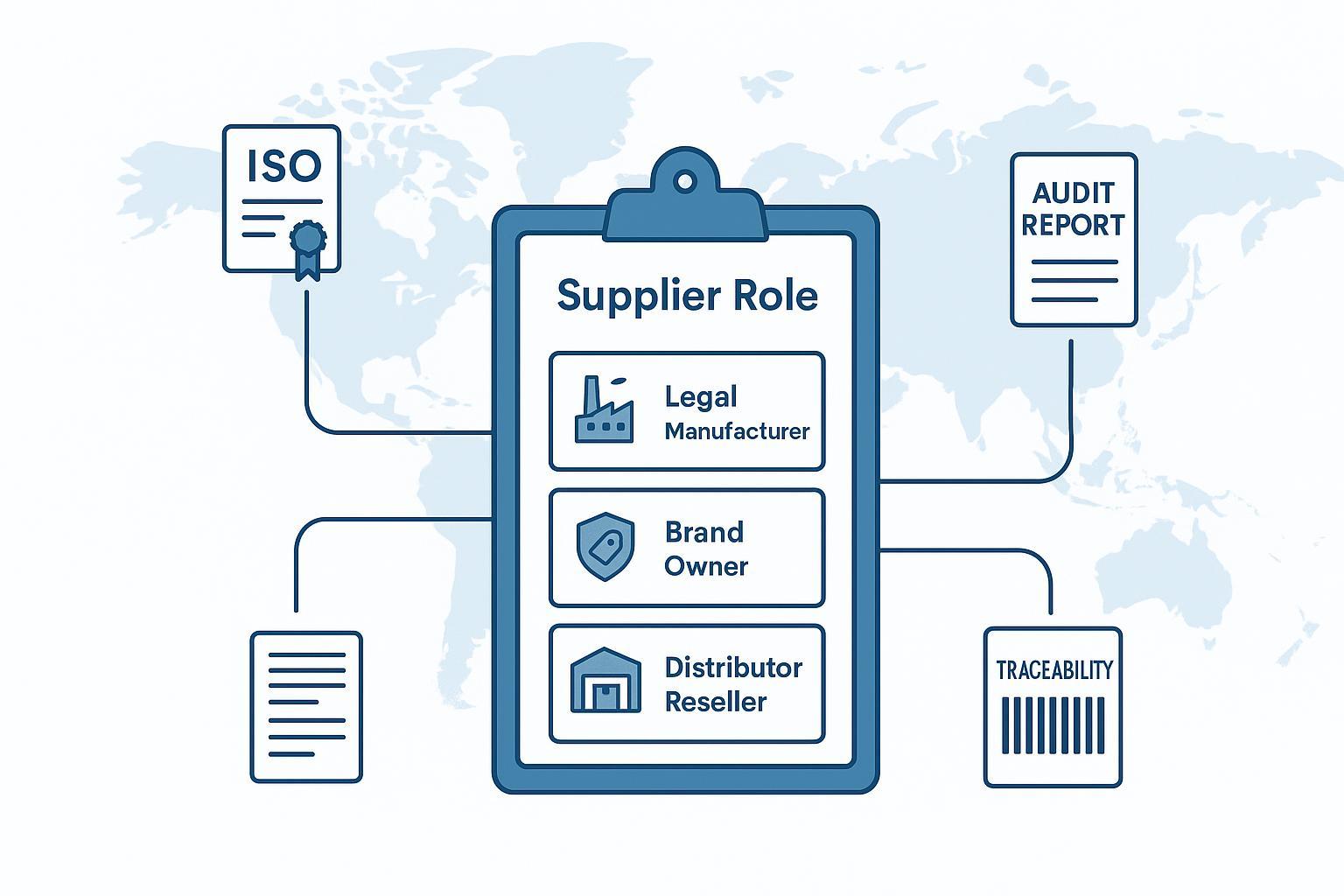
![Ilustrɛshɔn we de sho di ɔtpidik spɔlayt rol dɛn (ligal manifakta, brand ɔna, distribyushɔn) ɛn di dɔkyumentri-fɔs vetin flɔ]()
Pipul dɛn de yuz 'supplier' fɔ min difrɛn tin dɛn: wan manifakta, wan kɔntrakt manifakta, wan distribyushɔn, ɔ ivin wan tredin kɔmni. Dat ambiguity na risky.
If yu de bil wan shɔtlist fɔ orthopedic suppliers , di fɔs kwɛstyɔn simpul: Yu de evalyu di ligal manifakta, di brand ɔna, ɔ distribyushɔn/risɛla? Yu dokumɛnt chɛklist kin chenj dipen pan di ansa.
Dis na di pɔynt bak usay bɔku tim dɛn kin split dɛn sɔs len: sɔm kwɛstyɔn dɛn na bɔt ɔtpidik implant saplay dɛn (divays dɛn we dɔn dɔn), we ɔda wan dɛn na bɔt OEM/ODM patna dɛn we de sɔpɔt yu prayvet-lɛbul rodmap. Di vetting logic de ɔvalap—bɔt di dɔkyumɛnt ɛn rispɔnsibiliti dɛn nɔ di sem.
Wan prɛktikal lɔ: if wan spɔlayt nɔ ebul fɔ ɛksplen dɛn wok klia wan (manufakchɔ vs. ɔtorizayt distribyushɔn vs. OEM/ODM patna), slo ɛn gɛt klia bifo yu tɔk bɔt prayz.
Stej 1: Wetin fɔ aks fɔ bifo yu ivin aks fɔ sampul
Dis stej na fɔ pruv se di saplay na rial, kwalifay, ɛn rɛdi fɔ dɔkyumɛnt—bifo yu bɔn tɛm pan sampul lɔjistik.
1) Kwaliti sistɛm pruf (ɛn skɔp) .
Aks fɔ:
If yu de kɔmpia bɔku bid dɛn, notis klia wan if wan saplay na ISO 13485 ɔtpidik saplay (ɛn if di sɛtifiket skɔp mach di ɛksaktɔl prodak famili dɛn we yu de sɔs).
Di ɔdit stetmɛnt we dɛn jɔs gɛt (us bɔdi gi di sɛtifiket, ɛn we i dɔn) .
Wan ay-lɛv kwaliti ɔvaviu: kɔmplen handel, nɔ-kɔnfɔmɛns kɔntrol, kɔrɛkt akshɔn prɔses
Wetin mek dis impɔtant: ISO 13485 na wan pan di klia glob ɔl signal dɛn we de sho se wan manifakta de rul wan kwaliti sistɛm we gɛt dɔkyumɛnt. Fɔ kros-bɔda program (inklud LATAM), yu go stil nid makɛt-spɛsifi k pruf—so trit ISO 13485 as di beslayn, dɔn chɛk wetin yu target kɔntri dɛn nid fɔ rɛjista, ɔdit, ɛn kɔntinyu fɔ fala di lɔ.
2) Regulatory pathway pruf fɔ di spesifik prodak dɛm we yu de sɔs
'Dɛn dɔn klia/rɛjista dɛn prɔdak ya?' na wan blunt kwɛstyɔn. Bɛtɛ:
Fɔ ɛni prɔdak famili we yu plan fɔ bay, aks us divays klasifikeshɔn de aplay na yu target makit ɛn wetin dat min fɔ di rigyuletɔri path.
Rikwaym pruf fɔ di makit-spɛsifi k pruf fɔ di kɔntri dɛn we yu de sɛl insay (ɛgz., pruf fɔ rɛjista/notis, listin sɛtifiket, ɔtorizayt ripɔt/rɛjista ɔlda ditil, ɔ ɔda ɔfishal dɔkyumentri—i dipen pan di jɔrisdikshɔn).
Ad wan layn na yu intake fɔm we dɛn kɔl rigyuletɔri path verifyeshɔn ɛn yu nid ɔl tu (a) wan rɛjista/klirans rɛfrɛns fɔ da prodak famili de na yu target kɔntri dɛn, ɔ (b) di dɔkyumɛnt bies fɔ ɛni klem 'ɛksɛmpshɔn,' inklud di bɔda/limiteshɔn dɛn.
⚠️ Wonin : 'Exempt' nɔ ɛva min 'unregulated.' I kin min se wan patikyula prɛmaket stɛp nɔ nid fɔ da divays tayp de na da jɔrisdikshɔn de—ɔda kɔntrol dɛn stil de aplay.
3) Traceability ɛn labeling rɛdi
Aks fɔ mek dɛn tɔk klia wan bɔt:
Lot/serial traceability (aw dɛn de trak frɔm finish divays bak to raw matirial ɛn prɔses)
Labeling controls (aw dɛn kin gri fɔ chenj di lɛbl ɛn rilis) .
UDI rɛdi usay i apɔynt (aw dɛn de manej ɛn chɛk di yunik aydentifaya dɛn) .
If yu distribyushɔn insay bɔku ɔspitul sistɛm dɛn, trit UDI traceability mɛdikal divays dɛn lɛk wan nɔ-negoshiable kapabiliti: yu nid wan saplay we go ebul fɔ sɔpɔt traceability, fil akshɔn, ɛn dɔkyumentri rikwest dɛn we yu nɔ improvise.
Ivin if yu nɔ nid ful lɛbl paket na dis stej, yu want fɔ no se di pɔsin we de gi yu di tin gɛt wan kɔntrol sistɛm—nɔto las minit sprɛdshit.
4) Matirial ɛn manufakchurin kɔntrol (splay-rɛdi vɛshɔn)
Na awareness stej, yu nɔ nid ful tɛknikal dosya. Yu want fɔ gɛt ansa dɛn we dɛn kin kɔntrol to:
Wetin na di standad implant matirial dɛm we dɛn yuz (ɛgz., taytaniɔm alɔy, stenlɛs stiɛl, PEEK) ɛn aw dɛn de mentein di sɛt?
Wetin na di impɔtant tin dɛn we dɛn kin du fɔ mek tin dɛn (mashin, finish, klin) ɛn aw dɛn kin inspɛkt dɛn?
Us tɛst/inspekshɔn ripɔt dɛn kin gi fɔ ɛni batch ɔ fɔ ɛni ɔda?
If di saplay nɔ ebul fɔ diskrayb inspekshɔn chɛk-point ɔ nɔ ebul fɔ sheb ɛni sampul ripɔt, dat na signal.
5) Wan 'udat de du wetin' map fɔ OEM/ODM prɔjek dɛm
If yu de plan fɔ prayvet-lɛbul, layt kɔstɔmayshɔn, ɔ kɔ-divɛlɔpmɛnt, aks:
If yu nid dip prokyumɛnt fɔm fɔ OEM/ODM ɛvalueshɔn, XC Medico gɛt ditayl risɔs: Ultimate Gayd fɔ Ɔtpidik OEM & ODM Prokyumɛnt.
Stej 2: Wetin fɔ validet bifo yu fɔs bay ɔda
![What to validate before your first purchase order Wetin fɔ validet bifo yu fɔs bay ɔda]()
Stej 1 de tɛl yu se di spɔlayt fit fɔ tɛst. Stej 2 na fɔ mek shɔ se yu ebul fɔ skel witout sɔprayz.
1) Chenj kɔntrol: aw yu kin no we sɔntin chenj
Aks aw di pɔsin we de gi di tin dɛn de du:
Drawing/spec chenj dɛn
Di chenj we di pɔsin we de gi di tin dɛn kin chenj
Di chenj dɛn we de apin na di prɔses (ɛgz., fɔ trit di sɔfa, fɔ klin) .
Dɔn aks di prɛktikal kwɛstyɔn: 'Aw dɛn go notis wi, ɛn us dɔkyumɛnt wi go gɛt?'
2) Fɔ handle kɔmplen, CAPA, ɛn fil akshɔn rɛdi
Yu nɔ nid dɛn ful CAPA fayl. Yu nid fɔ:
Dɛn kɔmplen intake flɔ (wetin data dɛn kin kapchɔ) .
Aw dɛn kin invɛstigat di tin dɛn we nɔ gri wit di lɔ
Aw dɛn kin kɔmyuniket pɔtɛnɛshɛl fil ishu to patna dɛn
Spɔla dɛn we nɔ ebul fɔ diskrayb wan disiplin lɔp fɔ kɔmplen ɛn kɔrɛkt akshɔn dɛn at fɔ trɔst pan skel.
3) Di tin dɛn we yu de op fɔ pak/sterilayz—na if i rili impɔtant to wetin yu bay
Nɔto ɔl prɔdak dɛn de sɛl steril. Bɔt if yu de fɛn prɔdak dɛn we dɛn pak wit stɛriyl (ɔ yu de abop pan di pɔsin we de gi yu fɔ mek i gɛt stɛriyl barɛri intɛgriti), aks fɔ:
If sterile validation nɔr de pat pan yu skɔp, kip dis sɛkshɔn simpul ɛn dokumɛnt wetin yu ripɔtabl fɔ vs. wetin di spɔlayt ripɔtabl fɔ.
4) Logistics ɛn Incoterms we de mach yu risk tolerance
Aks fɔ:
Standart lida tɛm dɛn bay prodak famili
Bak ɔda kɔmyunikeshɔn ɛkspɛkteshɔn dɛn
Shipping terms (Incoterms) ɛn import sɔpɔt (kɔstɔm pepa wok, DDP opshɔn dɛn if dɛn gi am)
Dis na di say we 'great unit price' kin fel bɔku tɛm—bikɔs di rial kɔst kin sho pan dilɛys, pat pan shipmɛnt, ɛn riaktiv kɔmyunikeshɔn.
5) Portfolio fit: dɛn kin sɔpɔt yu ɔspitul kɔstɔma dɛn fɔ lɔng tɛm?
Brod pɔtfolio kɔvarej kin ridyus di spɔlayt sprawl (ɛn rɛjista ɛfɔt) ova tɛm.
If yu want fɔ kwik kwik wan sanity-check prodak breadth, yu kin kɔmpia di spɔlayt in kategori kɔvarej wit wetin yu de sɛl tide. Fɔ XC Medico in katalog-lɛvel ɔvaviu, si di Prodakt dɛn pej.
Rɛd flag dɛn we fɔ slo yu dɔŋ
Nɔto ɔl rɛd flag de diskwalifay, bɔt ɛni wan fɔ trig dip verifyeshɔn.
Sɛtifiket dɛn we nɔ gɛt skɔp, de we dɛn fɔ dɔn, ɔ bɔdi we dɛn gi
'FDA apruv' frayz we nɔ gɛt klia wan bɔt wetin rili klia, ɛksɛmpt, ɔ aplay
Vague traceability ansa ('wi kin trak am') witout diskraib aw
No dokumɛnt chenj kɔntrol ɛkspɛkteshɔn
Ovaprɔmis pan lida tɛm we nɔ gɛt klia invɛntari/prɔdakshɔn plan
Ligal rol we nɔ klia (manufakchɔ vs risayla) .
Usay XC Medico fit (ɛgzampul spɔlayt prɔfayl)
If yu want ɛgzampul bɔt aw wan spɔlayt kin prɛzɛnt dɛnsɛf agens di chɛklist we de ɔp, XC Medico de pozishɔn as ɔtpidik implant ɛn inschrumɛnt manifakta wit wan mɔlti-sistim pɔtfɔlio (spayn, trauma, jɔyn, spɔt mɛrɛsin, ɛksternal fiksatɔ, CMF) ɛn inschrumɛnt.
Dɛn kin tɔk bak bɔt di savis ɛn sɔpɔt we impɔtant to di distribyushɔn dɛn—dokumɛnt sɔpɔt, rɛjista matirial sɔpɔt na difrɛn makit dɛn, ɛn OEM/ODM opshɔn dɛn. Fɔ no mɔ bɔt da len de, luk XC Medico in OEM / ODM savis dɛn.
Dis pat nɔto rɛkɛmɔndeshɔn; na ɛgzampul fɔ di kayn klem dɛm we yu fɔ translet to verifyeshɔn kwɛshɔn dɛm.
Di nɛks tin dɛn we yu fɔ du
If yu de bil wan shɔtlist, di muv we yusful pas ɔl na fɔ standad yu spɔlayt-kwalifayeshɔn paket ɛn sɛn di sem riŋwe to ɛvri kandidet.
Wan simpul kit fɔ stat:
Stej 1 dok list (sɛti, skɔp, rol klia) .
Stej 2 kɔntrol list (chenj kɔntrol, kɔmplen/CAPA, lɔjistik) .
Wan pej kɔmpiashɔn grid fɔ skor di saplay dɛn ɔltɛm
Mɛdikal/rɛgyulatɔri disklɛmayshɔn: Dis atikul na fɔ jenɛral prokyumɛnt ɛdyukeshɔn ɛn nɔto mɛdikal ɔ ligal advays. Di tin dɛn we dɛn nid kin difrɛn bay di kayn prɔdak ɛn di say we dɛn de jɔj—vɛrify wit kwalifay rigyuletɔri/kwaliti pɔshɔnal dɛn.
FAQ we dɛn kin aks
Ɔl di ɔtpidik implant dɛn nid di sem rigyuletɔri aprɔval ɔlsay?
Nɔ, di rod dɛn we dɛn kin yuz fɔ mek di lɔ kin difrɛn bay di kɔntri ɛn di kayn divays dɛn. Kwalifayeshɔn fɔ bi bay di makit-spɛsifi k dɔkyumentri fɔ usay yu de sɛl (ɛn usay dɛn go yuz di prɔdak), nɔto pan wan-sayz-fit-ɔl klem frɔm ɛni saplay.
ISO 13485 naf fɔ kwalifay wan saplay?
Na strɔng statin signal, bɔt nɔto di wan ol stori. Yu stil nid fɔ evalyu traceability, chenj kɔntrol, kɔmplen/CAPA disiplin, ɛn (we i rili impɔtant) pak/sterilayz validɛshɔn ɛn lɔjistik rilaybiliti.
Wetin na di big mistek we distribyushɔn dɛn kin mek we dɛn de vet di spɔlayt dɛn?
Start wit prayz. Start wit rol klia ɛn dɔkyumentri rɛdi fɔs, dɔn tɛst sɛmpul, dɔn negoshiet tɛm wans yu dɔn ridyus di kɔmplians ɛn saplae risk.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu