
Key takeaways te pawh a awm
Academic prestige hian spine line chu low-risk angin a lantir thei a —chutih rualin, ngawi rengin distributor-te tan chuan pricing power, tight terms, leh margin hniam tak hmangin sumdawnna lamah a thawk thei lo a ni.
Tender tam zawkah chuan 'real' gate hmasa ber hi i supplier-in a publication list a ni lo. I bid package hian compliance readiness, traceability, leh consistent taka deliver theihna a finfiah em tih hi a ni.
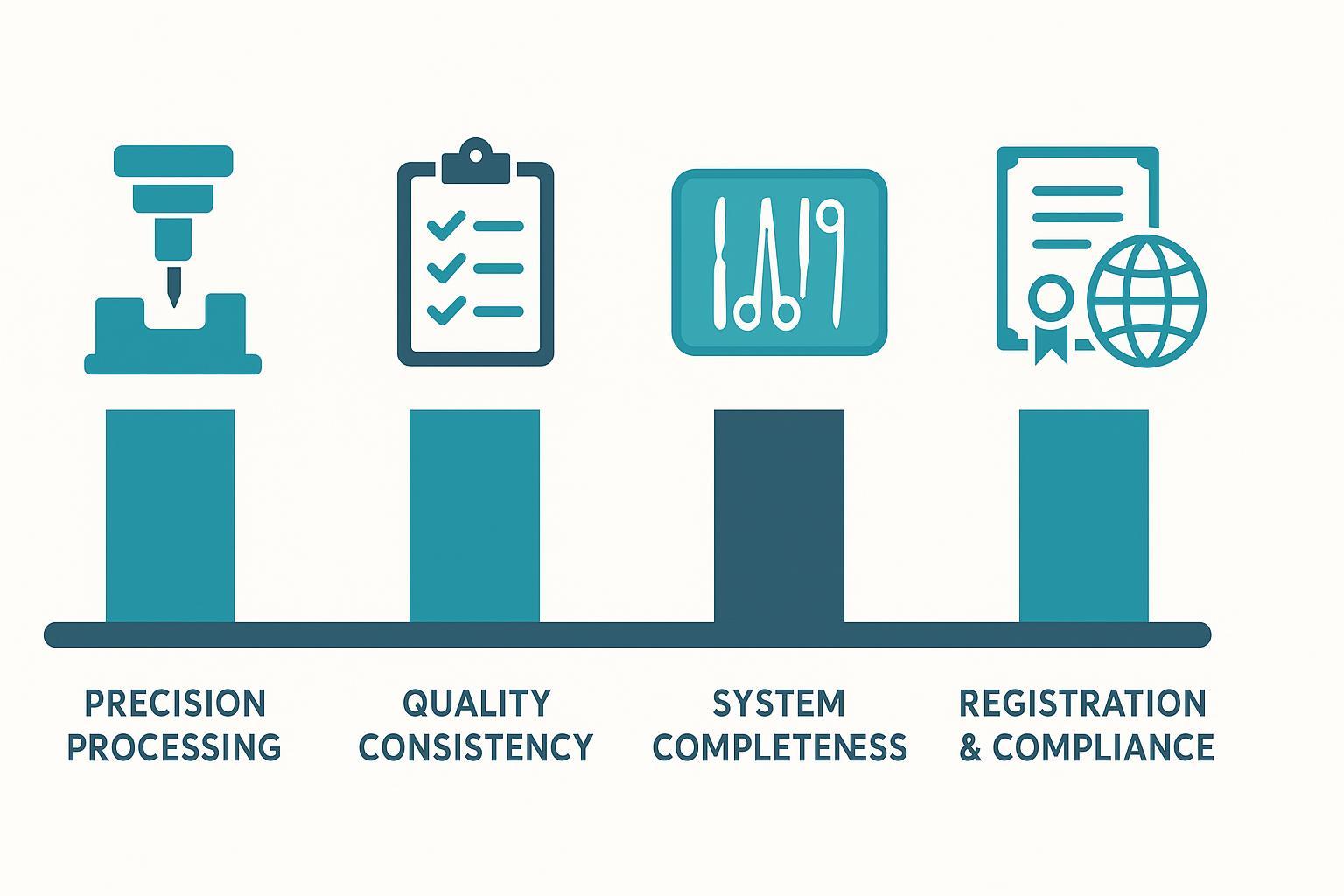
Bid hneh thei spine portfolio i duh chuan factory-te chu dimension pali-ah vet la: leh margin humhim thei precision processing , quality consistency , system completeness , leh registration/compliance experience.
Supplier inzawmna tha lo laka invenna kawng awlsam ber chu i audit theih tur evidence dil hi a ni: traceability entirnan, change-control posture, inspection methods, validated cleaning/sterilization pathways, leh instrument/tray logistics.
He movie hi i lo en tawh ngei ang.
'top-tier' spine brand i la a, a chhan chu clinical paper te, KOL slide deck te, leh instant credibility upgrade ang maia hriat hming hriat theihna chi hrang hrang a neih vang a ni. Tichuan sumdawnna chu kalpui i tum a:
Tender tamna hmunah chuan competitive takin i price thei lo.
I margin chu a squeeze a, chu chu basically handling fee a nih thlengin.
I contract chu 'partnership.' tia inthuam penalty set ang maiin a lang tan a.
Tin, a lungchhiatthlak ber chu: lehkhaah chuan mawhphurhna nei tak chu i ti a ni . 'evidence.' i thlang a.
Thutak semtute’n harsa taka an zir chhuah chu hetiang hi a ni:
Distribution economics tan chuan academic prestige hi thutlukna siamtu variable a ni fo lo. Factory siamna chakna chu a ni.
Paper-te chu a pawimawh loh vâng ni lovin— second-order filter an nih fo vâng a ni . First-order filter te hi boring tak tak an ni: documentation, consistency, completeness, leh mak tih loha scale-a execute theihna te.
The hidden mechanism: engvangin nge 'paper ropui tak tak' te hi distributor profit ah a chan loh
Brand lian te hian PDF an neih avangin an overcharging lo.
Market-in a phal avangin an overcharging a ni. Paper-te hi pricing power humhimtu thawnthua tel a ni—a bik takin hospital committee-te nena inven theih anga inhriat duhtute nen.
Distributor tan chuan chu pricing power chu predictable pattern angin a lang a:
Premium list man hi inbiakna apiangah baseline a lo ni ta a ni.
I 'discount' hi concession anga dah a ni a, sumdawnna kalphung nghet tak angin a awm lo.
Tender math hian hremna a pe che : market-in price-sensitive a neih poh leh chu premium chuan competitive bid-ah a block nasa zawk che a ni.
Risk gets pushed downstream : consignment terms, inventory liability, rigid minimums, leh performance clauses te hian adoption tiam aia slow a nih chuan bag chu i vawn tlat tir thin.
Chutih laiin i cost base chu implant chauh a ni lo.
A system chu a ni :
tray leh instrument hrang hrang a awm bawk
replenishment discipline neih a ni
documentation enkawl dan tur
ram hrang hrangah registration package pek a ni
labelling/UDI inpeih tawhna
lead time leh emergency replenishment te a ni
I supplier chuan factory-level repeatability hmanga chu piece te chu a execute thei lo a nih chuan distributor te chuan an pe thin—a tlangpuiin a man to berin: tender hlawhchham, stockout, leh 'urgent' logistics te.
Tender hian prestige an lei lo—proof an lei (spinal implant tender mamawh) .
Spine tender-ah chuan brand chak tak chuan thlamuanna siam turin a pui thei a ni. Mahse i submission hian gate a clear thei lo a nih chuan comfort hian bid a hneh lo.
Hospital RFP tak takte chu engtin nge an insiam dan hi han en teh: an thil phut tlangpui chu proposal chhanna kimchang, pricing exhibit, leh product scope zau tak an mamawh a—chuan vendor-te chu thil dangte bakah an chhanna leh an senso chungchangah an zirchiang bawk.
Spine implant tender frame dan grounded example i duh chuan primary documents like Cook County Health-in a ruhro (spinal implants) RFP emaw ECMC-in ruhro a siamah RFP a dah a ni . Scoring dik tak chu a inang lo hle a nih pawhin, pattern chu a inmil vek a ni: i bid package kimchang leh rintlakna aṭanga teh i ni—brand mythology-ah ni lovin.
Chu chu a trap chu a ni:
I evaluation hi academic paper atanga tan a nih chuan reassurance atan i optimize a ni.
I evaluation hi factory strength + compliance readiness atanga tan a nih chuan tender survival leh margin atan i optimize a ni.
Spinal implant tender mamawhna hnuaia dimension pali awmte
Spine portfolio i siam (or i siam thar leh) a nih chuan, hei hi evaluation frame a ni a, chu chuan tihsual man to tak tak a veng thei a ni.
1) Thil tih dan dik tak (precision processing) theihna
Spine implant hi siam chhuah a ngai hle. Surgeon preference card, instrument compatibility, leh tray replenishment standardize i tum hian variation tenau te chu operational problem lian tak a lo ni ta a ni.
Precision processing capability hi 'CNC an nei lo.' A ni:
geometries complex tak tak siamna tur multi-axis machining hmanga siam a ni
lot hrang hranga tolerances vawng tlat thei process stable
metrology capacity chu part complexity nena inmil a ni
factory hian production load tak tak hnuaiah result a repeat thei tih finfiahna
Eng nge dil tur (evidence, thutiam ni lovin):
Eng machining process nge in-house vs. outsourced?
Complex geometries (eg, CMM for dimensional verification) atan hian eng inspection tools nge hman?
Lot number nena inzawm representative inspection record an entir thei ang em?
Key Takeaway : Precision hi sumdawnna lam thil a ni. Inconsistency hi 'sample tha' chu warranty cost, tender risk, leh surgeon churn-a chantirtu a ni.
2) Quality inmilna (quality consistency) a ni
Distributor te hi batch tha lo pakhatah hrem an ni lo. An sawi lawk theih loh avangin hrem an ni.
Implant siamnaah quality consistency a awm tihna a ni a, supplier hian system a nei a, chu chuan variability a tihziaawm a, issue a man hmasa ber a ni:
in-process monitoring (final inspection chauh ni lovin) .
sampling plan tihfel leh traceable record te a awm bawk
a change-control mindset (chuvangin i 'same SKU' chu ngawi rengin product danglam takah a lo chang lo)
disciplined CAPA behavior (corrective and preventive action), a chhan chu issues a thleng thin—a pawimawh ber chu engtin nge an enkawl tih hi a ni
Hemi chungchang ngaihtuah dan awlsam tak:
3) System famkimna
Spine hi 'single implant' sumdawnna a ni tlem hle.
Hospital leh surgeon-te chuan system hrang hrang an zirchiang a: implant, instrument, tray, IFU, packaging, labeling, leh replenishment workflow. Chumi zinga pakhat tal a awm loh chuan i sales team chu operational friction vel ah hralhnaah an tang tlat a ni.
System kimchang takah hian:
coherent implant families (chuvangin incompatibilities i stitch khawm lo)
instrument/tray inpeih tawhna chu OR workflow tak tak atan
documentation kimchang (IFU, labeling, traceability beisei) 1.1.
i logistics siam thar leh ngai lovin procedure type hrang hranga scale theihna
Spine offering a zau theih dan tur concrete reference i duh chuan spine-system scope page like XC Medico Spine System , chu chuan spine instrument set hrang hrang leh implant chhungkaw hrang hrang a tarlang a ni. Thupui chu 'he catalog dik tak hi thlang rawh.' a ni lo. A thupui ber chu: tender-a hnehna changtu spine line chu system a ni a, SKU a ni lo.
4) Registration leh compliance lama tawnhriat nei (ISO 13485 certified manufacturer readiness) .
Tender leh ram hrang hranga sem chhuahnaah chuan compliance experience hi leverage (the good kind) a ni.
Khawvel pum huapa sem chhuah tura inpeih tak tak siamtu chuan:
regulatory document package hrang hrang a awm bawk
A tulna hmunah chuan stable labeling/UDI readiness a awm
traceability beisei a ni
audit posture (certificate, scope, leh renewal cadence chiang tak) .
Baseline atang hian tan la rawh: ISO 13485.
ISO 13485:2016 hi ISO-in a siam medical-device quality management system standard a ni—en rawh ISO hian ISO 13485:2016 standard a siam a ni . Distributor tan chuan a pawimawh a, a chhan chu qualification, audit sawihona, leh documentation dilna kalpui mekte inkara inhnialna a tihziaawm vang a ni.
Mahse, certificate-ah hian tawp mai suh.
I thil lei tur product family nena inmil scope leh evidence dil rawh. Certificate dik tak, implant siamna huam lo emaw, a kaihhnawih site leh process hrang hranga map lo emaw chu red flag a ni.
Spine implant supplier evaluation: 'paper chhiar,' chu titawp la, auditable proof dil tan rawh
Hetah hian due-diligence sequence a awm a, chu chuan tender leh real operation hnathawh dan a map a ni.
Step 1: Paper te hi atan hmang ang che secondary screen
Clinical evidence hian a pui thei a, chu chu:
design novel tak tak i buaipui a ni
hospital committee-te chuan thlamuanna atan zirlai chi hrang hrang an mamawh a ni
competitive account-ah surgeon adoption support i mamawh a ni
Mahse paper hi i kawngkhar hmasa ber a ni tur a ni lo—a chhan chu paper hian tender-in a hrem che zawhnate chu a chhang lo a ni:
Quarter leh lamah pawh hetiang quality hi i deliver thei ang em?
Thla tam tak chhunga back-and-forth awm lovin registration hi i support thei ang em?
Tray leh instrument te chu buaina awm lovin i replenish thei ang em?
Step 2: Factory-strength scorecard run hmasa phawt ang che
Factory tha tak chuan evaluation that’s specific chu a lawm hle ang.
'Quality sang tak i ni em?' tih zawh ai chuan artifacts zawt rawh:
lot number pakhat tan traceability entirnan
geometries complex tak takte enfiah dan tur
change-control process (engtin nge inthlak danglamnate chu inhrilhhriat leh pawm a nih?)
complaint chhui dan leh khar dan (CAPA posture) .
i target procedure te tan 'system completeness' chu eng ang nge a nih
Hei hi structure turin distributor-friendly framework i duh chuan XC Medico ang bawkin criteria-first approach hmang la Kum 2026-a Orthopaedic Supplier thlan dan tur Evaluation Criteria Top 7 —chumi hnuah chuan spine-specific realities angin siam rawh.
Step 3: Supply-chain execution chu validate (logistics-ah margin a thih avangin)
operational questions te zawt la : hmain Tender i thehluh hmasak ber
Standard lead time leh surge lead time te hi eng nge ni?
Instrument leh tray te replenishment process hi eng nge ni?
An warehousing leh inventory-control posture chu eng nge ni?
Hei hi 'factory strength' a lo lang ta a ni.
Entirnan, manufacturing-focused overview ang chi XC Medico-a Manufacturing Powerhouse hian process stage bik (machining, surface finishing, assembly, cleaning) a sawifiah a, ISO 13485 leh CE marking ang chi certification te nen '99.9% Inventory Accuracy,' ang chi operational claims te a tarlang bawk.
A thupui ber chu claims chu mitdel anga pawm loh hi a ni. a ni . auditable, operational language hmanga thusawi supplier te dah pawimawh hmasak Marketing language chauh ni lovin
Counterargument chak ber chu: 'Mahse clinical evidence erawh a pawimawh '.
A ti ve tho.
Thil tihsual chu clinical paper te hi thil dang zawng zawng proxy anga ngaih hi a ni.
Academic publication-te chuan:
hospital committee rintlak a ni
surgeon duhzawng sawihona
specific indication leh technique ngaihtuah turte
Mahse distributor-te tan chuan clinical evidence hi a thlaktu tur a ni lo:
registration peih fel tawh a ni
thil siam chhuah dan (consistent manufacturing) a ni
system kimchang tak tak a awm
supply sawi lawk theih a ni
Rilru thianghlim model chu hei hi a ni:
Orthopaedic OEM/ODM manufacturer shortlist i siam a nih chuan he inthliarna hi a pawimawh a ni—a chhan chu first filter dik lo chuan economics dik lo takah a lock che a ni.
Adoption comfort chauh i lei chuan a tawpah chuan business risk i pe ang.
Step dang: he reframe hi supplier shortlist ah chantir rawh
He thuziak hi nuam lo taka dik nia i hriat chuan, chu chu chhinchhiahna ṭha tak a ni—chu thang chu chiang takin i hmu a ni.
Hetah hian low-friction next step a awm: phek khata sei 'factory strength' checklist siam la, i ngaihtuah mek spine supplier zawng zawngah apply rawh.
A bul tanna tangkai tak chu XC Medico ang chi documentation-first vetting framework hi a ni practical guide to vetting orthopedic suppliers in the US , chutah chuan i target market leh tender realities-ah te insiamrem rawh.
Academic prestige atana premium pek ai chuan, distributor thang chak tak takte chuan orthopaedic supplier te nen thawhhona an nei a, precision processing leh proven compliance experience te chu an dah pawimawh ber a ni—i margin humhim chungin quality-a tender i hneh theih nan.
FAQ
1) 'Published clinical papers' hian spine tender-a ka chak theih nan min pui em?
An pui thei a, mahse i kawngkhar hmasa ber angin an pui thei lo.
Tender-heavy market tam zawkah chuan thuchhuah chuan thlamuanna (committee rinna, surgeon inbiakna) a thlawp a ni. Bid package cleartu chu compliance readiness, traceability, leh consistent supply execution proof a ni . Paper-te chu secondary screen angin en rawh—chumi hnuah auditable artifact hmangin factory chakna chu validate rawh.
2) Distributor margin squeeze thei tur spine supplier hmuh awlsam ber chu eng nge ni?
Risk downstream tirtu sumdawnna structure zawng rawh.
Warning sign hman tlanglawn ber chu tender math tih theih lohna premium list man, rigid minimum, inventory liability, leh penalty-heavy 'partnership' clause te a ni. Supplier-in pricing power a neih chuan handling-fee margin a hnutchhiah che a nih chuan, line chu sumdawnna lamah a chhe thei lo—brand chu a hmingthang tak angin lang mah se.
3) Spine line-a ka commit hmain eng document emaw evidence emaw nge ka dil tur?
I audit theih tur evidence dil la, i verify theih loh thutiam zawt lovin.
Lot number tak tak nena inzawm traceability entirnan, complex geometries-te tana representative inspection records, leh change-control approach chiang tak (inthlak danglamnate enfiah dan, pawm dan leh inbiakpawhna) hmangin tan la rawh. Tichuan CAPA posture leh complaint closure entirnan quality-system behavior te chu validate la, chubakah i target procedure nena inmil instrument/tray logistics te pawh i validate bawk.
4) Engtin nge 'system completeness' hi tender leh adoption nghawng tak tak tur angin ka evaluate ang?
System chu OR workflow angin evaluate la, catalog angin evaluate rawh.
Implant chauh i lei lo—tray, instrument, replenishment discipline, labeling/UDI readiness, IFUs, leh constant workaround awm lovin procedure type hrang hranga scale theihna i lei a ni. System kimchang leh inzawm tlat chuan surgeon-te tan friction a tihziaawm a, i team-te chu operational surprise tlem zawk hmanga bid thianghlim zawk thehluh turin a pui bawk.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu





