![ROI equation and supply chain icons illustrating total lifecycle cost when evaluating orthopedic suppliers]()
Most distributor teams don’t miscalculate ROI because they’re bad at math.
They miscalculate it because they’re using the wrong unit of analysis.
If you’re evaluating orthopedic suppliers by first-order margin (your gross margin on the first PO), you’re not measuring ROI. You’re measuring a single snapshot of unit economics—while the risks that actually make or break distributor profitability live in the months that follow.
Key Takeaway: A supplier can look “high ROI” on the first invoice and still be a negative-ROI partner once you account for compliance friction, instrument downtime, stockouts, and the cost of fixing problems.
This article is a thought-leadership reset for orthopedic device distributors: what ROI should mean in your world, where your “hidden denominator” lives, and a practical way to evaluate suppliers without fake precision.
First-order margin is not ROI (and why smart teams still fall for it)
First-order margin is seductive because it’s clean:
But ROI is a return on an investment over time. And supplier decisions are not one-time transactions—they’re operational commitments.
If your supplier line becomes a growth engine, you win repeatedly: repeat cases, additional hospitals, portfolio expansion, smoother tenders.
If your supplier line becomes a risk engine, you lose repeatedly: delays, documentation gaps, instrument problems, angry surgeons, emergency freight, rework, and—worst—lost accounts.
The trap is confusing what is easy to measure with what matters most.
What ROI should mean for orthopedic distributors
For distributors, the “investment” isn’t only the purchase price. It’s the total cost of partnership.
And the “return” isn’t only the first-order gross margin. It’s the total lifecycle cash generated from a supplier line that stays stable long enough to scale.
Here’s the practical reframe:
The two-part correction
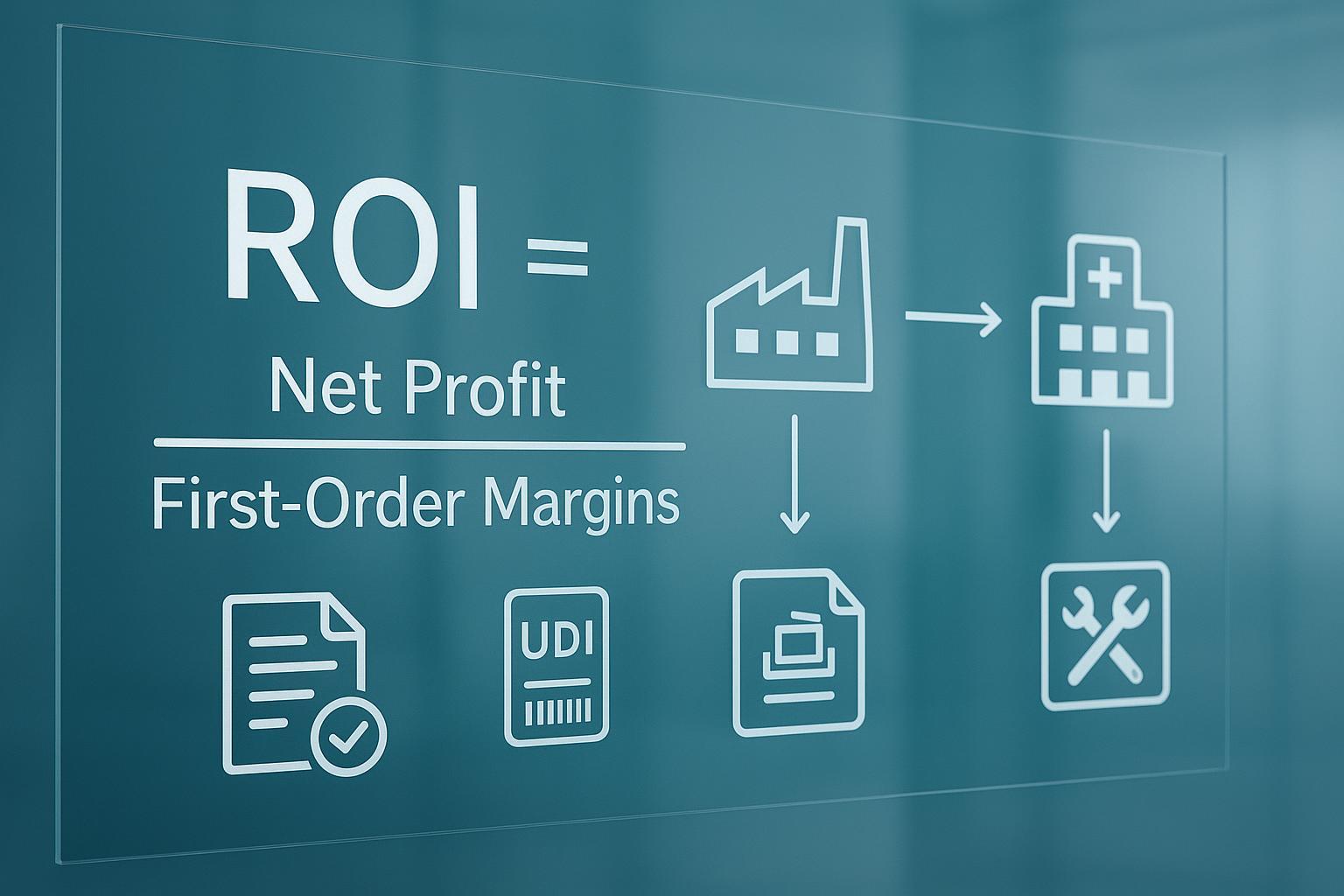
The numerator must be lifecycle revenue, not first-order margin.
The denominator must be lifecycle cost, not unit price.
In other words:
True ROI = (Total stable repeat revenue across channels over the product lifecycle) / (Total cost of the partnership)
That denominator is where most “cheap” suppliers become expensive.
The hidden denominator: where “cheap” suppliers create expensive outcomes
When a supplier’s offer looks too good, it’s usually because their quote is only pricing what’s visible—and leaving you to price what’s inevitable.
Below are three hidden-cost buckets that matter disproportionally in orthopedics.
1) Compliance friction: when missing documentation becomes a paid delay
Distributors don’t just move product. You move product through systems: registrations, tenders, hospital vendor onboarding, labeling requirements, traceability expectations.
When your supplier lacks the right documentation maturity, you don’t simply “wait a bit longer.” You pay—often in the form of:
delayed registrations or tender submissions
stalled hospital onboarding
extra rounds of clarification, rework, and translations
inventory you can’t sell yet (working capital tied up)
A common flashpoint is UDI (Unique Device Identification). In the US, FDA’s UDI system requires device identifiers to appear on labels and packages and requires device information submission to the Global Unique Device Identification Database (GUDID). The FDA explains the system and its requirements in UDI Basics and details policy timelines in UDI compliance policies and compliance dates.
The distributor lesson isn’t “UDI is hard.” It’s this:
⚠️ Warning: If your supplier can’t reliably produce the labeling/traceability artifacts your market expects, your timeline becomes the cost—and the cost is rarely in the quote.
Even if your target market isn’t the US, the same pattern holds: weak documentation maturity turns into schedule risk, and schedule risk turns into cash loss.
2) Instrument set reliability: when kit quality becomes throughput loss
Orthopedics is not just implants. It’s implants + instruments, and instruments behave like a shared asset.
If your instrument sets are inconsistent, fragile, or hard to maintain, the cost shows up as:
more frequent repairs and replacements
longer turnaround times between cases n- reduced set utilization (you need more sets to support the same case volume)
emergency courier shipments to move sets between hospitals
Distributors often model instruments as a one-time purchase (capex mindset). Operationally, it’s closer to a throughput constraint.
If sets are down for repair, your “ROI” isn’t a number. It’s a missed case.
A simple question exposes this risk quickly:
If the supplier can’t speak in operational terms—repair turnaround, spare-part availability, failure modes, and what “normal” looks like—you’re buying uncertainty.
3) Stockouts: when OTD failures become surgeon churn
Stockouts don’t just create backorders.
In orthopedic distribution, stockouts can cause:
cancelled or delayed surgeries
surgeons trying (and then sticking with) an alternative system
hospital procurement losing confidence in your reliability
tender risk (“we can’t award if supply isn’t stable”)
This is where the ROI illusion turns brutal: one missed delivery can erase months of margin.
Operationally, the relevant metric isn’t whether the supplier “tries their best.” It’s whether they reliably hit ship dates.
On-time delivery (OTD) is a standard supply chain reliability KPI—commonly measured as the share of orders delivered on or before the promised date. For a representative definition and context, see Geotab’s “On-time delivery” glossary entry (2025).
In distributor reality, OTD is not a logistics metric. It’s a surgeon-retention metric.
Reconstruct the ROI formula (the version finance will respect)
Here’s a distributor-ready formula that avoids fake precision while forcing the right thinking.
The ROI equation
True ROI = (Total lifecycle revenue from stable repeat purchasing) / (Total lifecycle cost of partnership)
The numerator: total lifecycle revenue
Include revenue that depends on supplier stability:
repeat cases from existing accounts
expansion into additional hospitals/surgical centers
portfolio pull-through (e.g., trauma line helps you win spine; spine opens doors for joint)
tender renewals enabled by reliable performance
The key word is stable. Revenue that only exists in best-case assumptions shouldn’t be counted the same as revenue supported by operational evidence.
The denominator: total lifecycle cost
At minimum, model:
Purchase price (unit price, instruments, freight, duties, payment terms impact)
Communication cost (time across procurement, QA/RA, operations, sales support)
Compliance sunk cost (documentation rework, regulatory delays, audit preparation)
After-sales / maintenance cost (instrument repairs, spare parts, replacements, field issues)
Stockout cost (expedites, lost cases, account churn risk)
This aligns with the broader procurement discipline of evaluating total cost of ownership. ISM outlines the lifecycle mindset in Understanding Total Cost of Ownership in Procurement, and also notes how ROI metrics can be misleading if the denominator is artificially minimized in The Monthly Metric: Procurement ROI.
A 30-minute ROI worksheet (no spreadsheets required)
You don’t need a complex model to avoid the illusion. You need a consistent set of inputs.
Use this lightweight worksheet to compare Supplier A vs Supplier B.
Step 1: Write down your “operational truth window”
Pick a realistic period—e.g., 12 months.
enough time for repeats, service events, and documentation cycles to show up
short enough that your assumptions are still defensible
Step 2: Estimate stable revenue (conservative)
Ask your commercial team:
How many accounts will actually adopt this line in 12 months?
What’s a conservative monthly case volume per account?
What’s the average revenue per case?
Then apply a stability haircut if you have delivery/documentation uncertainty.
Step 3: Price the hidden denominator explicitly
For each supplier, add line items:
Documentation cycle time: How many rounds do you expect? Who is involved? (RA, QA, sales ops)
Instrument set downtime risk: What’s the repair lead time? Spare availability?
Delivery reliability: What happens when the supplier misses ship date? Do you expedite? Do you lose cases?
Even rough numbers are better than ignoring the categories.
Step 4: Compare outcomes, not just quotes
A supplier that is 8% cheaper on unit price can still be 30% more expensive in total cost if:
you miss one tender window due to documentation gaps
one instrument set is out of rotation for weeks
one high-volume surgeon switches due to a stockout
Your model doesn’t need to be perfect. It needs to be complete.
The diligence questions that reveal real ROI
If you want to scale a medical device distribution business sustainably, you must shift your focus from unit price to total lifecycle profitability.
Evaluate international orthopedic suppliers based on their ability to reduce your total cost through reliable on-time delivery, robust QA systems, and localized logistics.
Here are questions that force real answers:
Delivery and availability
What is your OTD/OTIF performance over the last 6–12 months?
What are standard lead times by product family, and what are the exception paths?
What is your backorder policy and allocation policy during demand spikes?
Documentation and compliance
Which certificates and scopes are current and easily shareable (ISO 13485, CE where applicable, etc.)?
What is your change notification process (materials, design, suppliers, labeling)?
How do you support traceability/UDI requirements where relevant?
Instruments and after-sales
What are the common failure modes for instrument sets?
What is your repair turnaround SLA, and what spare parts are stocked?
Do you provide maintenance guidance and recommended lifecycle replacement schedules?
Communication and escalation
What is the escalation path when a shipment is late or documentation is blocked?
Who owns the problem: sales, operations, or QA/RA?
If a supplier can’t answer these with specifics, you’re not comparing suppliers—you’re comparing stories.
What “good” looks like (without making this a vendor pitch)
Awareness-stage content shouldn’t end with “therefore buy from us.”
But it should end with a clearer picture of what to look for.
A credible supplier partner is one who can show:
You’re not buying “cheap implants.” You’re buying a supply system that either protects or destroys your downstream revenue.
FAQ: ROI, TCO, and supplier evaluation for distributors
Is first-order margin useless?
No—it’s just incomplete.
First-order margin is a quick signal of unit economics. The mistake is treating it as the whole story. A supplier can look strong on the first PO and still destroy profitability through delays, rework, instrument downtime, or stockouts.
What’s the difference between ROI and total cost of ownership (TCO)?
Think of TCO as the cost side of the ROI equation.
TCO: all-in lifecycle cost (purchase price + operating/maintenance + risk/friction costs).
ROI: lifecycle return relative to lifecycle cost (your total stable cash generated divided by total lifecycle cost).
If you only track unit price, you’re tracking a small subset of TCO.
Which hidden cost is usually the biggest for distributors?
In orthopedics, it’s often stockouts and timeline slip—because they damage surgeon trust and hospital continuity.
A single missed case can erase the savings you negotiated on dozens of items.
What documents should we request before we place the first PO?
Ask for a distributor-ready packet that matches your target market requirements. At a minimum, most teams request:
current quality management certificates (scope matters, not just the logo)
product labeling and traceability approach (including UDI where applicable)
change notification and complaint/CAPA process overview
IFU and product labeling samples for review
For UDI context in the US market, the FDA provides a clear overview in its UDI basics guidance (linked earlier in this article).
OTD vs OTIF: which one should we use?
Use both if you can.
OTD (On-time delivery) tells you whether shipments arrive when promised.
OTIF (On-time, in-full) tells you whether you receive the full order on time.
If a supplier is “on time” but routinely short-ships, your downstream operations still break.
How do we price the cost of a stockout without guessing?
Don’t try to model everything. Price one conservative scenario:
pick one high-volume account
estimate the revenue of one missed case
add a conservative probability of churn if stockouts recur
Even rough scenario pricing is better than implicitly pricing stockouts at $0.
When does it make sense to switch suppliers—even if the margin looks good?
When the lifecycle denominator is expanding faster than your numerator.
Common signals:
recurring documentation blocks that delay registrations/tenders
instrument sets repeatedly out of rotation
missed ship dates that create surgeon complaints or hospital escalation
How long should we evaluate a new supplier before calling the ROI “real”?
A practical window is 6–12 months—long enough for delivery patterns, documentation cycles, and after-sales responsiveness to reveal themselves.
Next steps
If you want, I can share a one-page distributor checklist that maps:
the ROI formula inputs
the hidden-cost categories to price
the documentation and QA artifacts to request upfront
the OTD/OTIF questions that reveal whether a supplier is built to scale
Use it to pressure-test every new supplier conversation—before the first PO makes your ROI look great on paper.
Русский
English
简体中文
繁體中文
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
አማርኛ
ພາສາລາວ
ភាសាខ្មែរ
Bahasa Melayu
ဗမာစာ
தமிழ்
Filipino
Bahasa Indonesia
magyar
Română
Čeština
Монгол
қазақ
Српски
हिन्दी
فارسی
Kiswahili
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Latine
Dansk
اردو
Shqip
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
isiXhosa
ייִדיש
Yorùbá
isiZulu